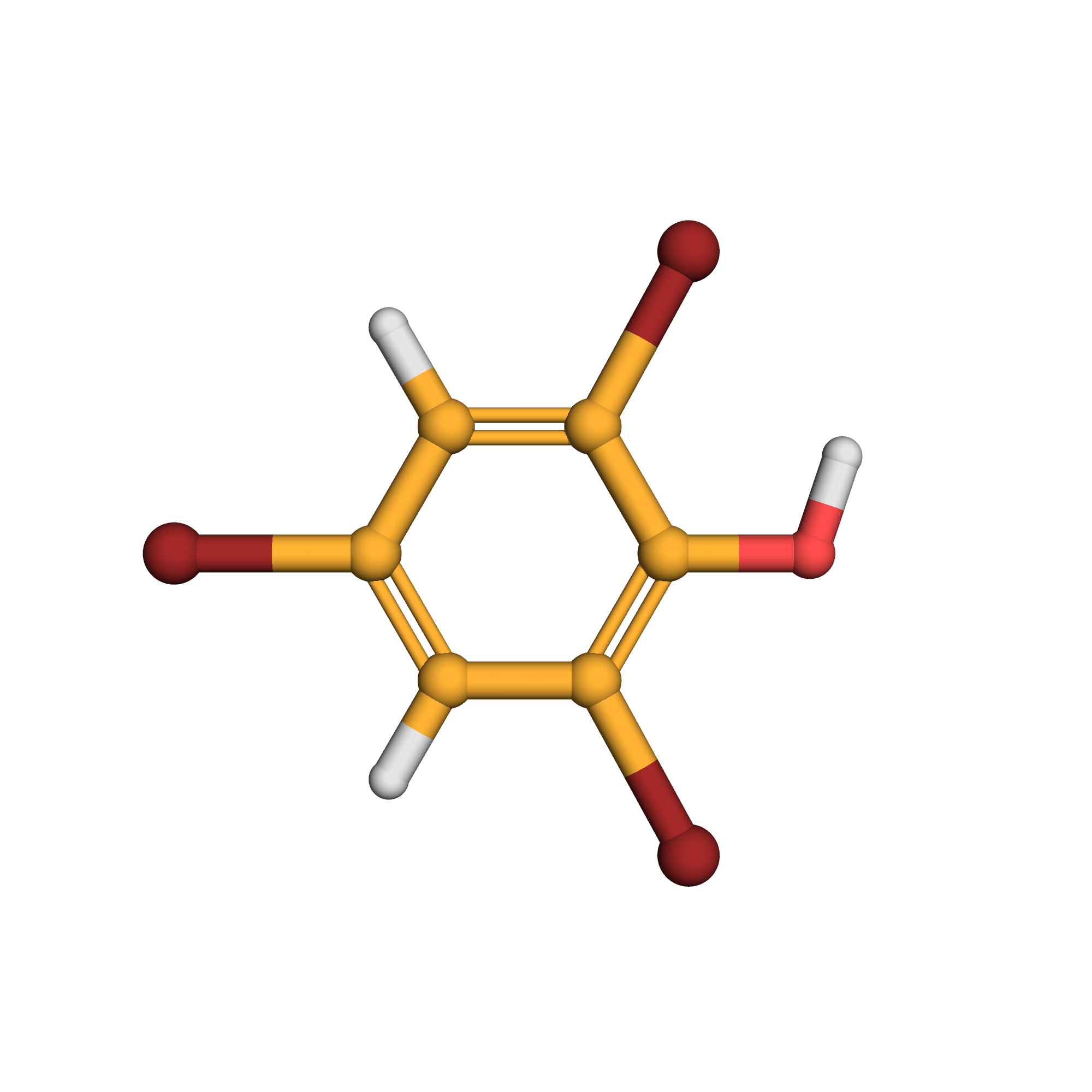
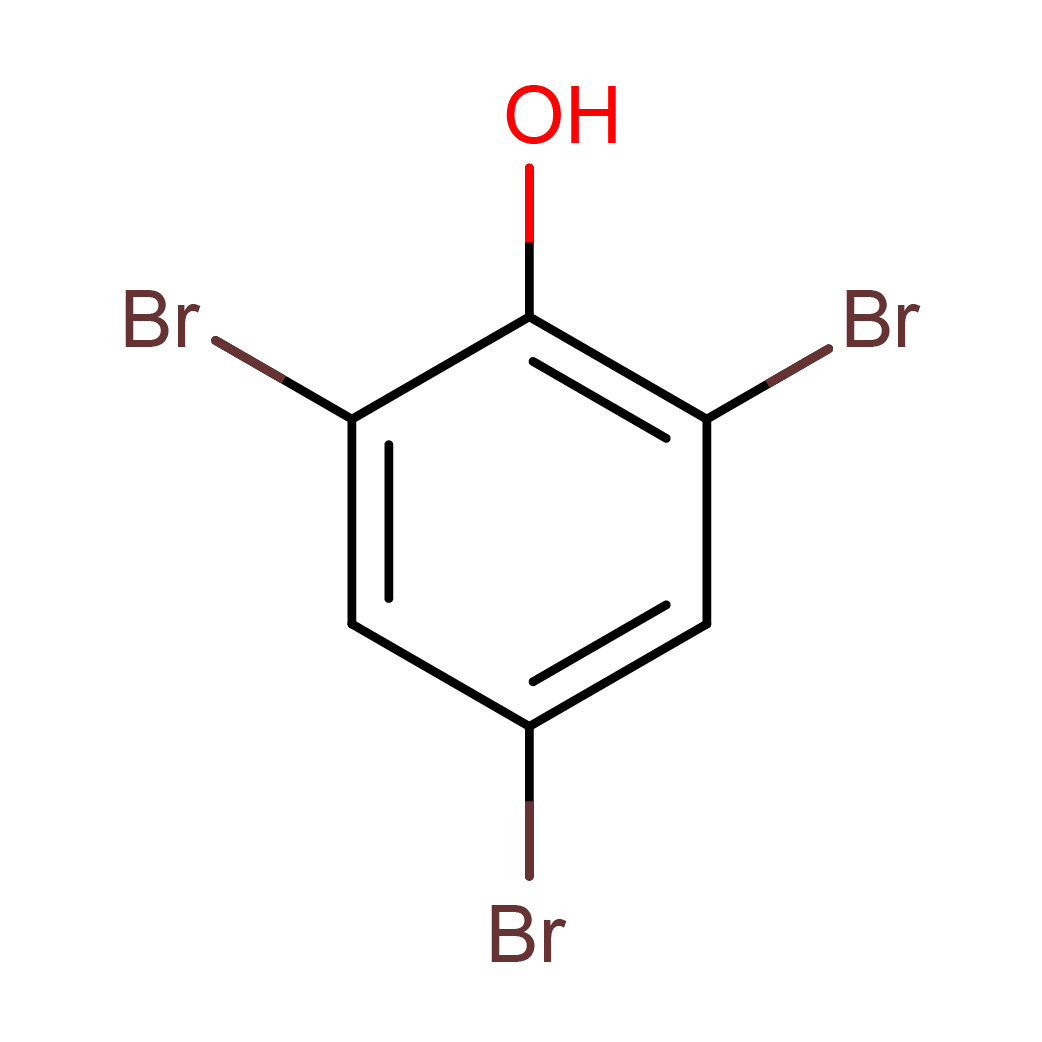
2,4,6-tribromophenol
Synonyms: "2,4,6-tribromophenol", "tribromophenol", "Bromol", "Bromkal pur 3", "Xeroform", "Flammex 3BP", "2,4,6-TBP".
Source: 2,4,6-Tribromophenol is used also as an flame retardant in epoxy, polyurethane, plastics, paper, textiles and others.
Identifiers:
IUPAC Name: 2,4,6-tribromophenol
CAS Number: 118-79-6
PubChem ID: 1483
InChiKey: BSWWXRFVMJHFBN-UHFFFAOYSA-N
Canonical SMILES: C1=C(C=C(C(=C1Br)O)Br)Br
Structural Properties:
Molecular Formula: C6H3Br3O
Molecular Weight: 330.801
Pharmacophore Features:
Number of bond donors: 1
Number of bond acceptors: 1
Number of atoms different from hydrogen: 10
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Hamers T, Kamstra JH, Sonneveld E, Murk AJ, Kester MH, Andersson PL, Legler J, Brouwer A. 2006. In vitro profiling of the endocrine-disrupting potency of brominated flame retardants. Toxicol Sci 92(1):157-173. DOI: 10.1093/toxsci/kfj187. URL: https://academic.oup.com/toxsci/article/92/1/157/1642946Meerts IATM, van Zanden JJ, Luijks EAC, van Leeuwen-Bol I, Marsh G, Jakobsson E, Bergman a?, Brouwer A. 2000. Potent competitive interactions of some brominated flame retardants and related compounds with human transthyretin in vitro. Toxicol Sci 56(1):95-104. DOI: 10.1093/toxsci/56.1.95. URL: https://academic.oup.com/toxsci/article/56/1/95/1646061.
External Links



2D-structure
3D-structure