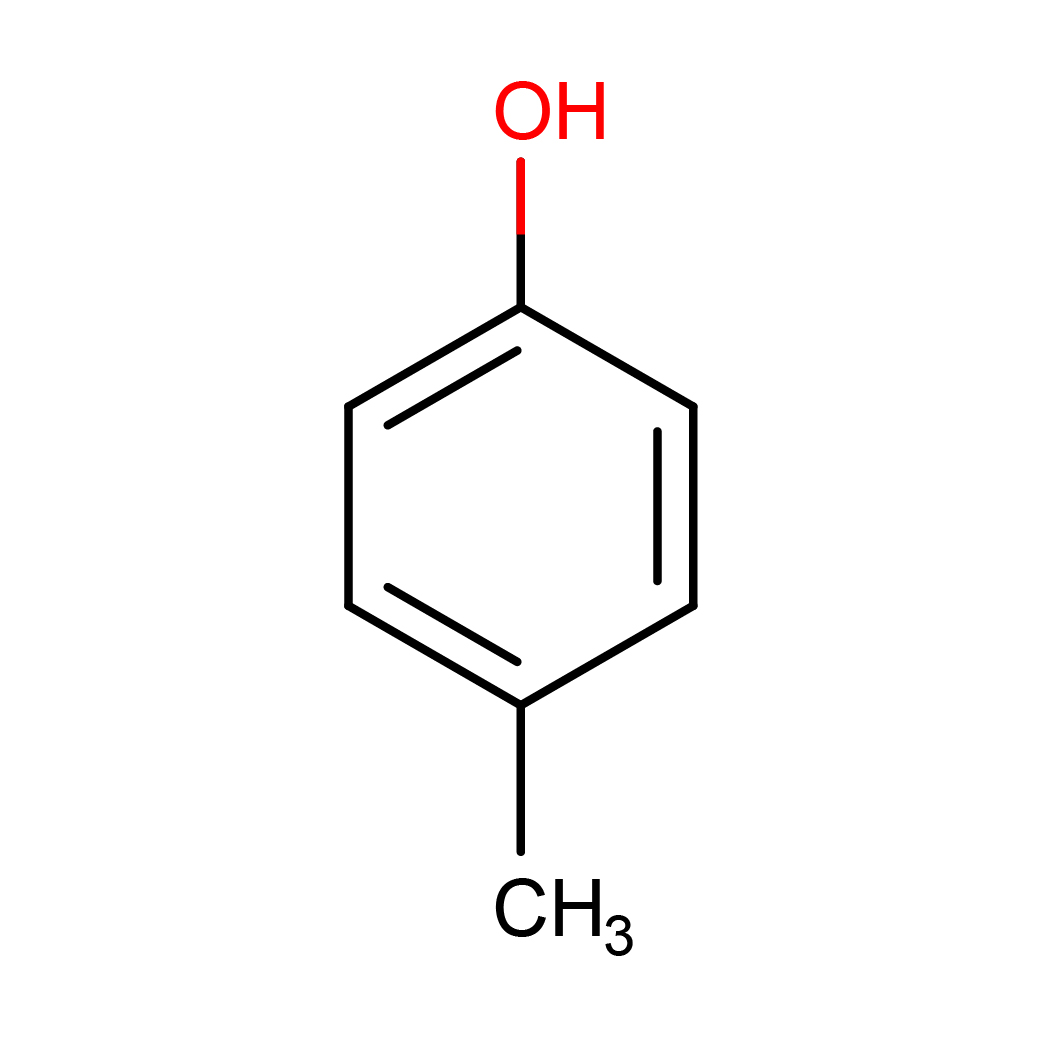
p-cresol
Synonyms: "4-cresol", "4-methylphenol", "4-hydroxytoluene", "p-methylphenol", "p-hydroxytoluene", "p-tolyl alcohol", "p-oxytoluene", "para-cresol"
Source: p-cresol is used in disinfectants and fumigants, in the manufacture of synthetic resins, photographic developers and explosives.
Identifiers:
IUPAC Name: 4-methylphenol
CAS Number: 106-44-5
PubChem ID: 2879
InChiKey: IWDCLRJOBJJRNH-UHFFFAOYSA-N
Canonical SMILES: CC1=CC=C(C=C1)O
Structural Properties:
Molecular Formula: C7H8O
Molecular Weight: 108.138
Pharmacophore Features:
Number of bond donors: 1
Number of bond acceptors: 1
Number of atoms different from hydrogen: 8
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Kawakami K, Makino I, Kato I, Uchida K, Onoue M. 2009. p-Cresol inhibits IL-12 production by murine macrophages stimulated with bacterial immunostimulant. Immunopharmacol Immunotoxicol 31(2):304-309.
Nakamura M, Yamazaki I, Kotani T, Ohtaki S. 1989. Thyroglobulin-mediated one- and two-electron oxidations of glutathione and ascorbate in thyroid peroxidase systems. J Biol Chem 264(22):12909-12913.
Nishihara T, Nishikawa J, Kanayama T, Dakeyama F, Saito K, Imagawa M, Takatori S, Kitagawa Y, Hori S, Utsumi H. 2000. Estrogenic activities of 517 chemicals by yeast two-hybrid assay. Journal of Health Science 46(4):282-298.
Thompson DC, Perera K, Fisher R, Brendel K. 1994. Cresol isomers: comparison of toxic potency in rat liver slices. Toxicol Appl Pharmacol 125(1):51-58.
External Links



2D-structure
3D-structure