dibenz[a,h]anthracene
Synonyms: "dibenz[a,h]anthracene", "dibenz(A,H)anthracene", "1,2,5,6-dibenzanthracene", "dibenzo[a,h]anthracene", "dibenzo(a,h)anthracene", "dibenz(a,h)antracene", "1,2:5,6-Dibenzoanthracene".
Source: Dibenz[a,h]anthracene is ubiquitously present in the environment as a minor component of the total content of polycyclic aromatic hydrocarbons mainly arising from incomplete combustion of carbonaceous material.
Identifiers:
IUPAC Name: naphtho[1,2-b]phenanthrene
CAS Number: 53-70-3
PubChem ID: 5889
InChiKey: LHRCREOYAASXPZ-UHFFFAOYSA-N
Canonical SMILES: C1=CC=C2C(=C1)C=CC3=CC4=C(C=CC5=CC=CC=C54)C=C32
Structural Properties:
Molecular Formula: C22H14
Molecular Weight: 278.354
Pharmacophore Features:
Number of bond donors: 0
Number of bond acceptors: 0
Number of atoms different from hydrogen: 22
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Machala M, Vondracek J, Blaha L, Ciganek M, Neca JV. 2001. Aryl hydrocarbon receptor-mediated activity of mutagenic polycyclic aromatic hydrocarbons determined using in vitro reporter gene assay. Mutat Res 497(1-2):49-62. DOI: 10.1016/S1383-5718(01)00240-6. URL: https://www.sciencedirect.com/science/article/pii/S1383571801002406.
Vinggaard AM, Hnida C, Larsen JC. 2000. Environmental polycyclic aromatic hydrocarbons affect androgen receptor activation in vitro. Toxicology 145(2-3):173-183. DOI: 10.1016/S0300-483X(00)00143-8. URL: https://www.sciencedirect.com/science/article/pii/S0300483X00001438.
External Links


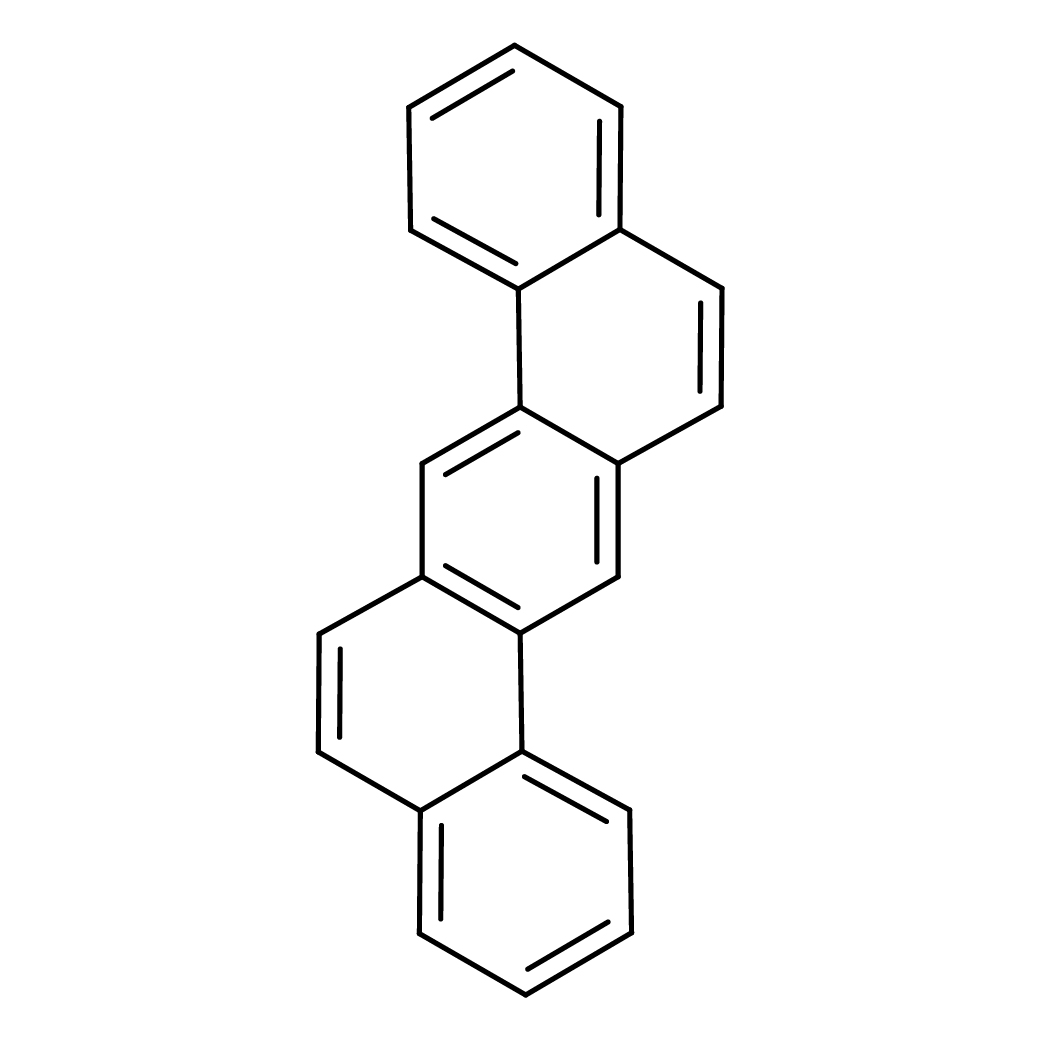
2D-structure
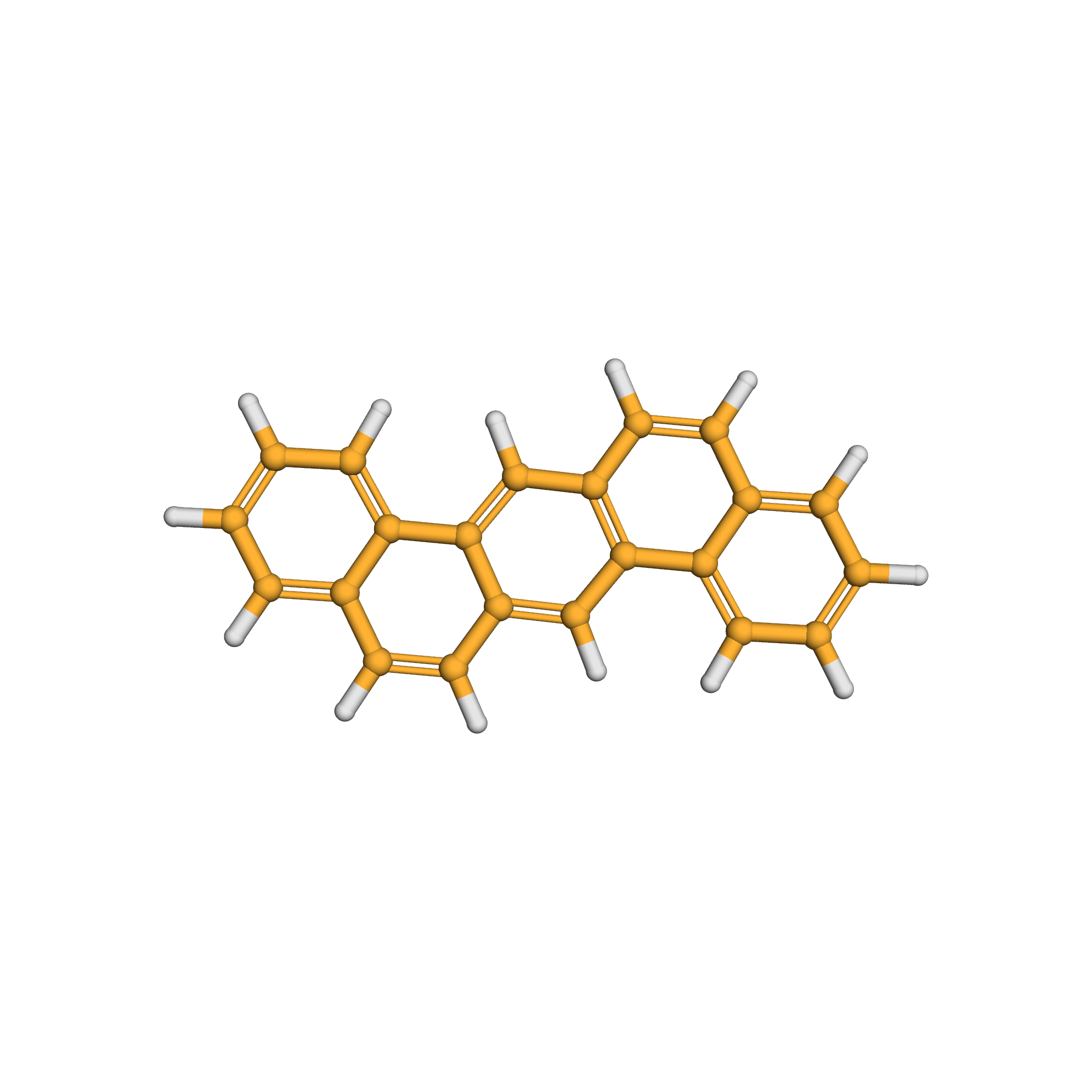
3D-structure