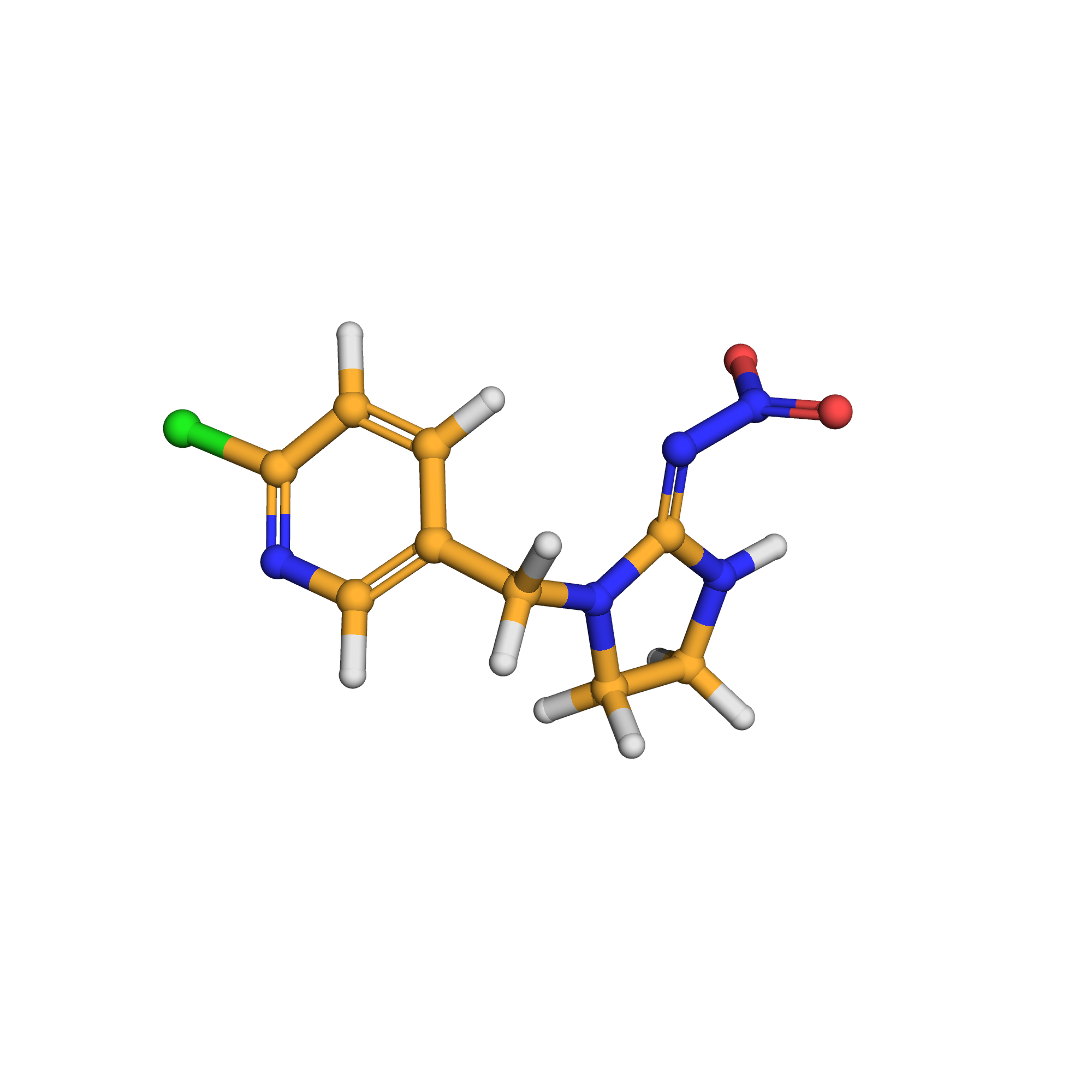
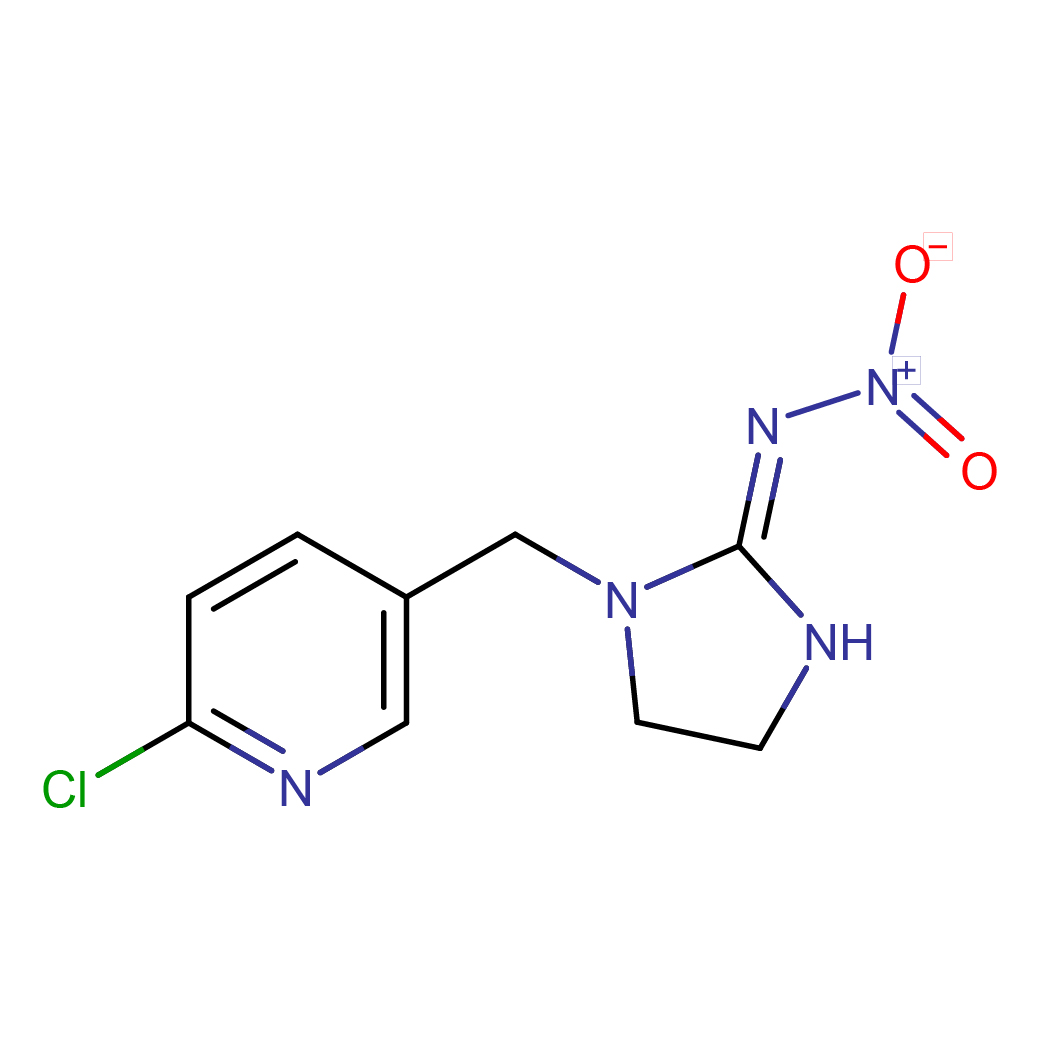
1-((6-chloro-3-pyridinyl)methyl)-N-nitro-2-imidazolidinimine
Synonyms: "(2E)-1-[(6-chloropyridin-3-yl)methyl]-N-nitroimidazolidin-2-imine", "1-((6-Chloro-3-pyridyl)methyl)-N-nitro-2-imidazolidinimine", "1-((6-Chloro-3-pyridinyl)methyl)-N-nitro-2-imidazolidinimine", "(E)-imidacloprid", "(2Z)-1-[(6-chloropyridin-3-yl)methyl]-N-nitroimidazolidin-2-imine", "Premise 75", "Provado", "Confidor", "Admire", "Gaucho", "Merit", "Confidor SL", "Confidor 200 SL", "Imidaclopride".
Source: Confidor is a systemic insecticide.
Identifiers:
IUPAC Name: (NE)-N-[1-[(6-chloropyridin-3-yl)methyl]imidazolidin-2-ylidene]nitramide
CAS Number: 138261-41-3
PubChem ID: 86287518
InChiKey: YWTYJOPNNQFBPC-UHFFFAOYSA-N
Canonical SMILES: C1CN(C(=N[N+](=O)[O-])N1)CC2=CN=C(C=C2)Cl
Structural Properties:
Molecular Formula: C9H10ClN5O2
Molecular Weight: 255.662
Pharmacophore Features:
Number of bond donors: 1
Number of bond acceptors: 4
Number of atoms different from hydrogen: 17
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Bal R, Naziroglu M, Turk G, Yilmaz O, Kuloglu T, Etem E, Baydas G. 2012 Aug. Insecticide imidacloprid induces morphological and DNA damage through oxidative toxicity on the reproductive organs of developing male rats. Cell Biochem Funct 30(6):492-9. DOI: 10.1002/cbf.2826. URL: http://onlinelibrary.wiley.com/doi/10.1002/cbf.2826/abstract.
Caron-Beaudoin, E, Viau, R, Hudon-Thibeault, AA, Vaillancourt, C, and Sanderson, JT. 2017. The use of a unique co-culture model of fetoplacental steroidogenesis as a screening tool for endocrine disruptors: The effects of neonicotinoids on aromatase activity and hormone production. Toxicology and applied pharmacology 332:15-24. DOI: 10.1016/j.taap.2017.07.018. URL: https://www.sciencedirect.com/science/article/pii/S0041008X17303150.
Kapoor U, Srivastava MK, Srivastava LP. 2011. Toxicological impact of technical imidacloprid on ovarian morphology, hormones and antioxidant enzymes in female rats. Food Chem Toxicol 49(12):3086-3089. DOI: 10.1016/j.fct.2011.09.009. URL: https://www.sciencedirect.com/science/article/pii/S0278691511004522?via%3Dihub.
Kim J, Park Y, Yoon KS, Clark JM, Park Y. 2013. Imidacloprid, a neonicotinoid insecticide, induces insulin resistance. J Toxicol Sci 38(5):655-60. DOI: 10.2131/jts.38.655. URL: https://www.jstage.jst.go.jp/article/jts/38/5/38_655/_articleKugathas S, Audouze K, Ermler S, Orton F, Rosivatz E, Scholze M, Kortenkamp A. 2015 (in press). Effects of common pesticides on prostaglandin D2 (PGD2) inhibition in SC5 mouse Sertoli cells, evidence of binding at the COX2 active site, and implications for endocrine disruption. Environ Health Perspect. DOI: 10.1289/ehp.1409544. URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4829986/.
Najafi G, Razi M, Hoshyar A, Shahmohamadloo S, Feyzi S. 2010. The Effect of Chronic Exposure with Imidacloprid Insecticide on Fertility in Mature Male Rats. International Journal of Fertility and Sterility 4(1):9-16. URL: http://ijfs.ir/journal/article/abstract/2392Park Y, Kim Y, Kim J, Yoon KS, Clark J, Lee J, Park Y. 2013. Imidacloprid, a neonicotinoid insecticide, potentiates adipogenesis in 3T3-L1 adipocytes. J Agric Food Chem 61(1):255-259. DOI: 10.1021/jf3039814. URL: http://pubs.acs.org/doi/10.1021/jf3039814.
External Links

2D-structure
3D-structure