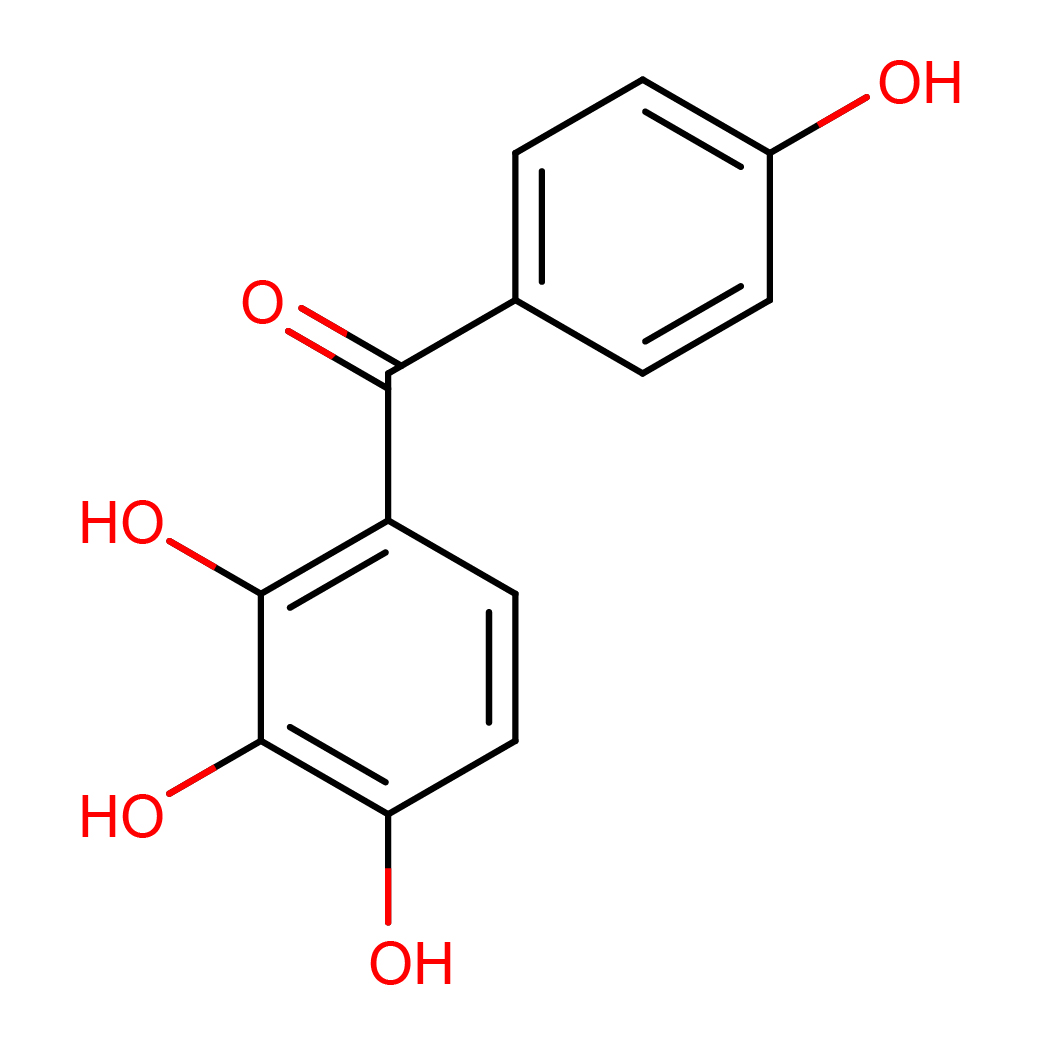
2,3,4,4'-tetrahydroxybenzophenone
Synonyms: "(4-hydroxyphenyl)(2,3,4-trihydroxyphenyl)methanone", "2,3,4,4'-tetrahydroxy benzophenone".
Source: 2,3,4,4'-tetrahydroxybenzophenone is used in the synthesis of photosensitive resins.
Identifiers:
IUPAC Name: (4-hydroxyphenyl)-(2,3,4-trihydroxyphenyl)methanone
CAS Number: 31127-54-5
PubChem ID: 919792
InChiKey: ZRDYULMDEGRWRC-UHFFFAOYSA-N
Canonical SMILES: C1=CC(=CC=C1C(=O)C2=C(C(=C(C=C2)O)O)O)O
Structural Properties:
Molecular Formula: C13H10O5
Molecular Weight: 246.218
Pharmacophore Features:
Number of bond donors: 4
Number of bond acceptors: 5
Number of atoms different from hydrogen: 18
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Kawamura Y, Mutsuga M, Kato T, Iida M, Tanamoto K. 2005. Estrogenic and anti-androgenic activities of benzophenones in human estrogen and androgen receptor mediated mammalian reporter gene assays. Journal of Health Science 51(1):48-54. DOI: 10.1248/jhs.51.48. URL: https://www.jstage.jst.go.jp/article/jhs/51/1/51_1_48/_article.
Kawamura Y, Ogawa Y, Nishimura T, Kikuchi Y, Nishikawa J, Nishihara T, Tanamoto K. 2003. Estrogenic activities of UV stabilizers used in food contact plastics and benzophenone derivatives tested by the yeast two-hybrid assay. Journal of Health Science 49(3):205-212. DOI: 10.1248/jhs.49.205. URL: https://www.jstage.jst.go.jp/article/jhs/49/3/49_3_205/_article.
External Links


2D-structure
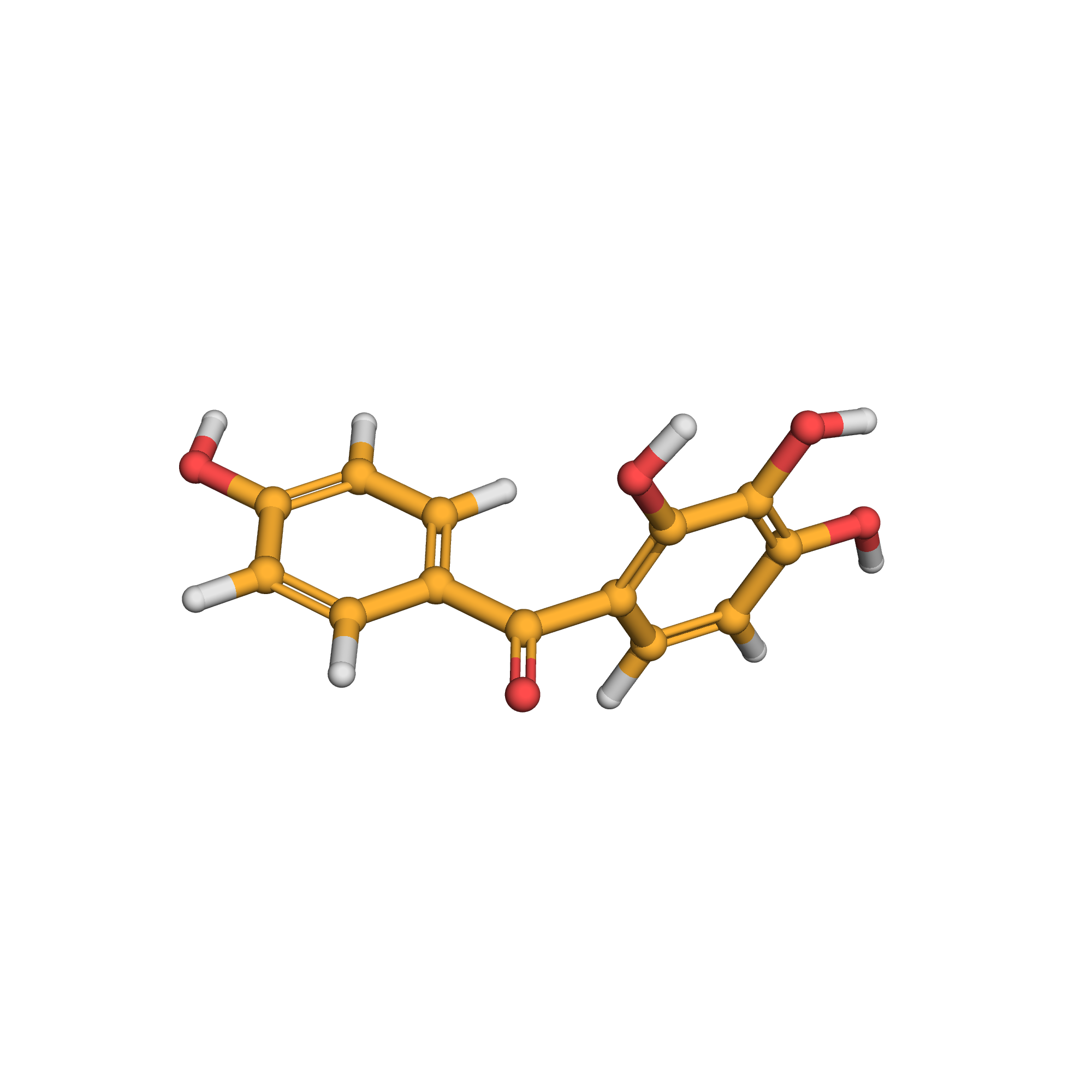
3D-structure