malathion
Synonyms: "carbofos", "carbophos", "karbofos", "mercaptothion", "phosphothion", "cythion", "prioderm", "sadophos", "carbetox"
Source: malathion is an organophosphate insecticide commonly used to control mosquitos and other flying insects.
Identifiers:
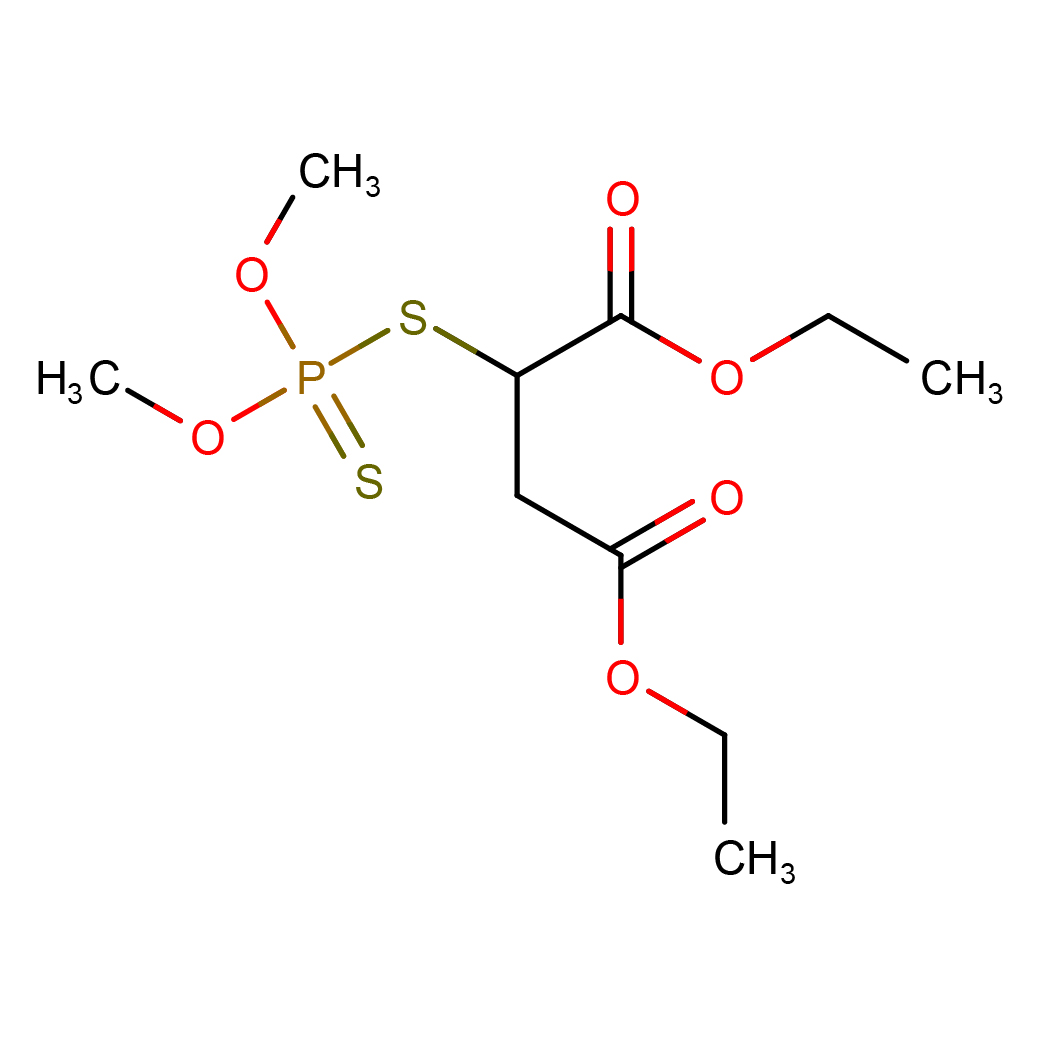
IUPAC Name: diethyl 2-dimethoxyphosphinothioylsulfanylbutanedioate
CAS Number: 121-75-5
PubChem ID: 4004
InChiKey: JXSJBGJIGXNWCI-UHFFFAOYSA-N
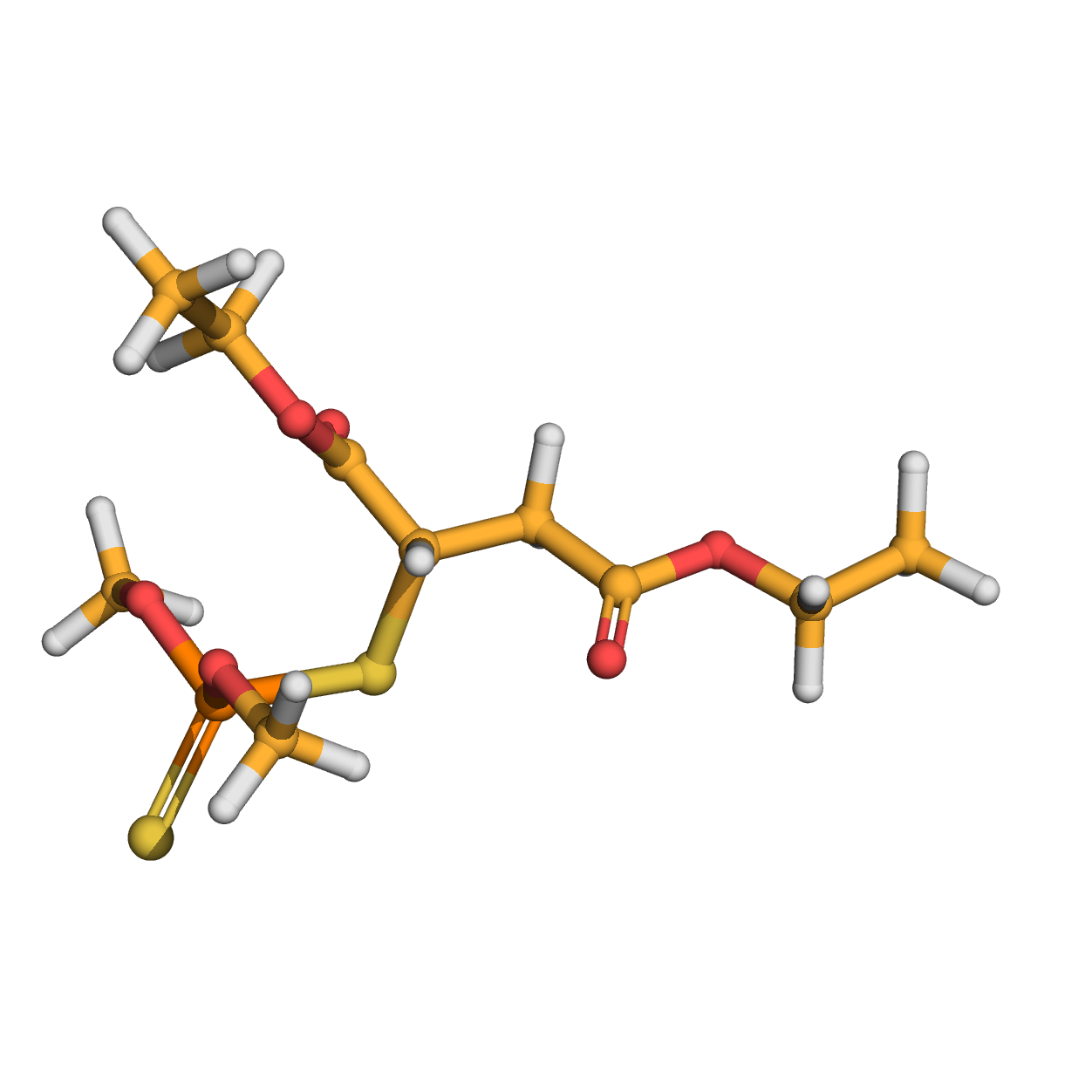
Canonical SMILES: CCOC(=O)CC(C(=O)OCC)SP(=S)(OC)OC
Structural Properties:
Molecular Formula: C10H19O6PS2
Molecular Weight: 330.358
Pharmacophore Features:
Number of bond donors: 0
Number of bond acceptors: 2
Number of atoms different from hydrogen: 19
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Ozmen G, Akay MT. 1993. The effects of malathion on some hormone levels and tissues secreting these hormones in rats. Veterinary & Human Toxicology 35(1):22-24.
Panahi P, Vosough-Ghanbari S, Pournourmohammadi S, Ostad SN, Nikfar S, Minaie B, Abdollahi M. 2006. Stimulatory effects of malathion on the key enzymes activities of insulin secretion in Langerhans islets, glutamate dehydrogenase and glucokinase. Toxicology Mechanisms & Methods 16(4):161-167.
Pournourmohammadi S, Farzami B, Ostad SN, Azizi E, Abdollahi M. 2005. Effects of malathion subchronic exposure on rat skeletal muscle glucose metabolism. Environmental Toxicology & Pharmacology 19(1):191-196.
Reuber MD. 1985. Carcinogenicity and toxicity of malathion and malaoxon. Environ Res 37(1):119-153.
van den Berg KJ, van Raaij JAGM, Bragt PC, Notten WRF. 1991. Interactions of halogenated industrial chemicals with transthyretin and effects on thyroid hormone levels in vivo. Arch Toxicol 65(1):15-19.
External Links

2D-structure
3D-structure