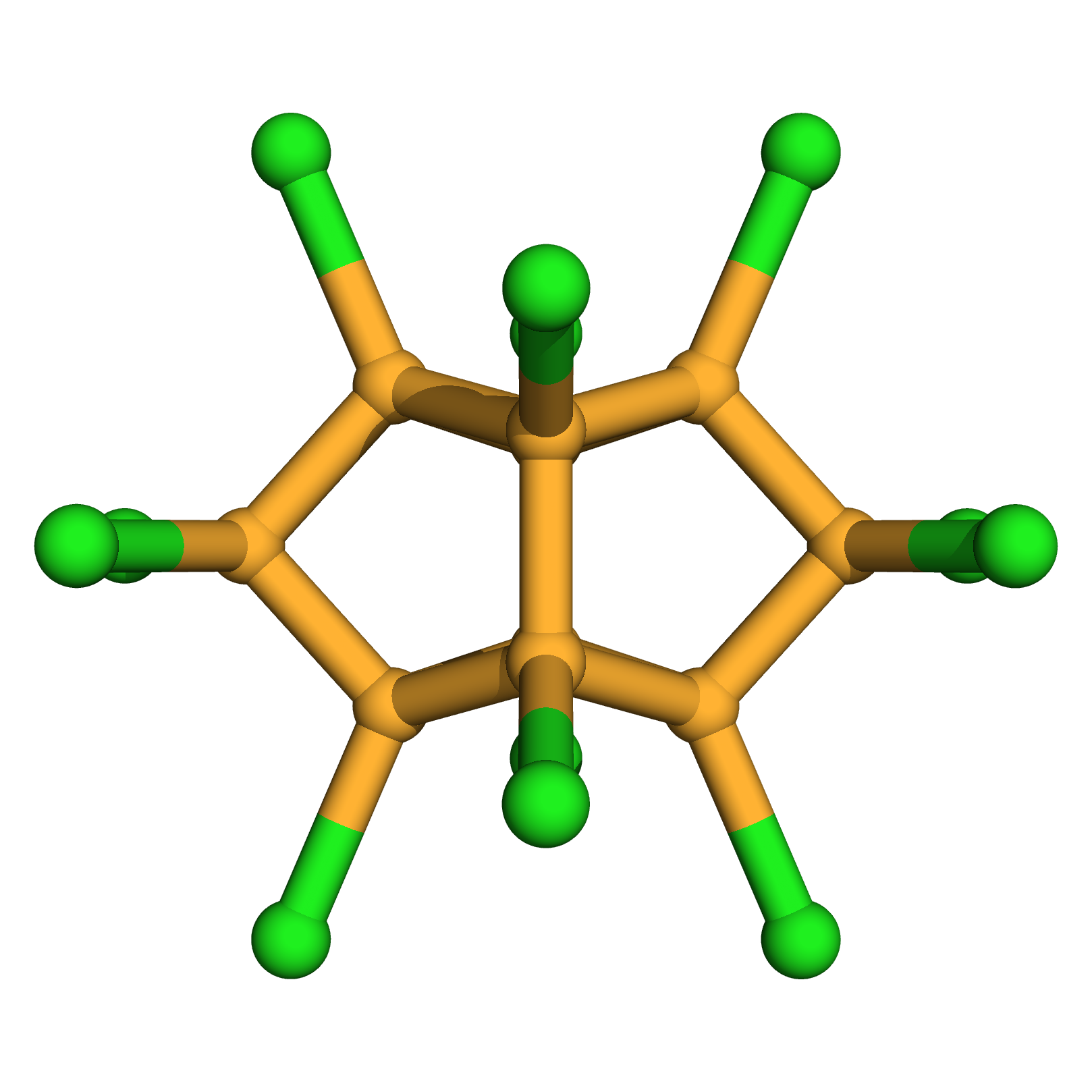
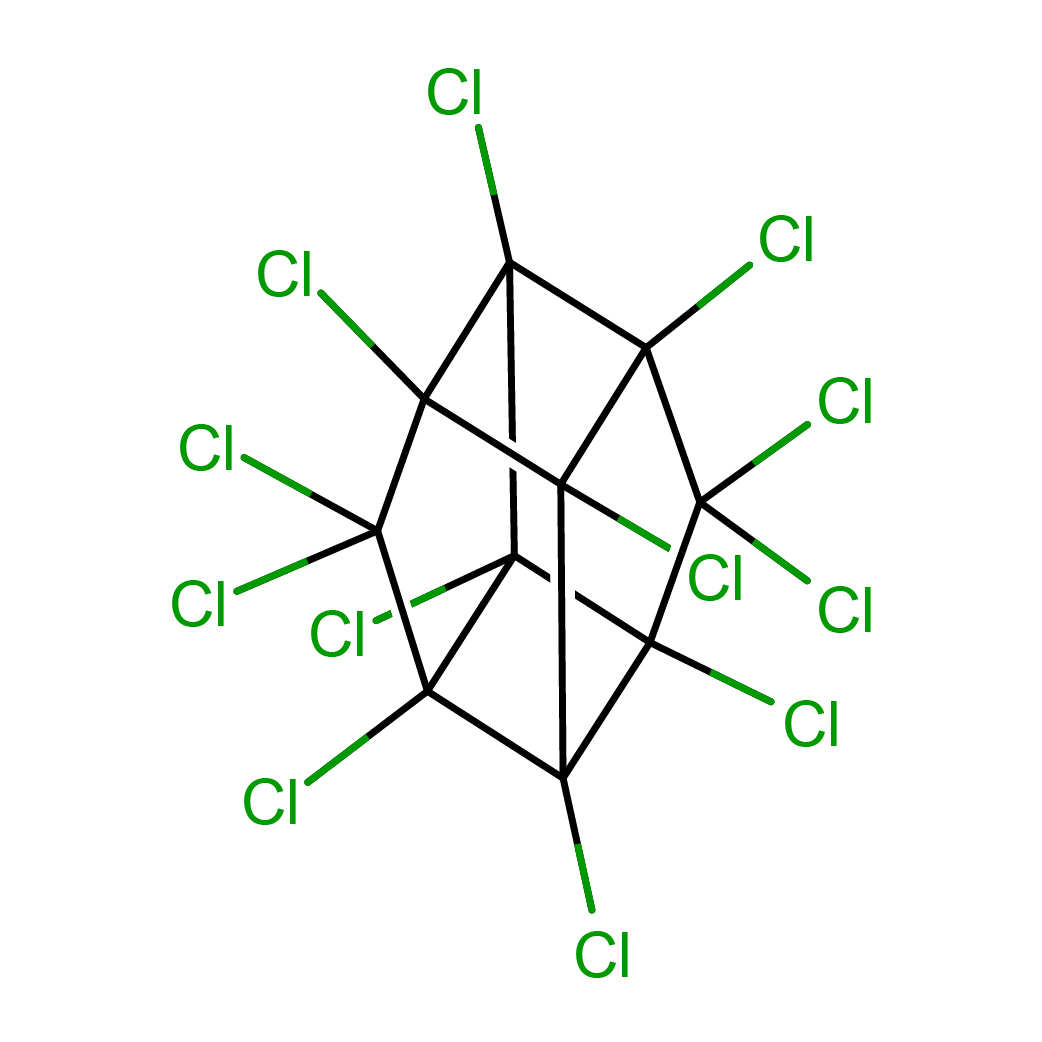
mirex
Synonyms: "dechlorane", "perchloropentacyclodecane", "paramex", "perchlorodihomocubane", "bichlorendo", "dodecaclor", "ferriamicide", "perchlordecone"
Source: mirex is a bait insecticide used against a number of insect pests.
Identifiers:
IUPAC Name: 1,2,3,4,5,5,6,7,8,9,10,10-dodecachloropentacyclo[5.3.0.02,6.03,9.04,8]decane
CAS Number: 2385-85-5
PubChem ID: 16945
InChiKey: GVYLCNUFSHDAAW-UHFFFAOYSA-N
Canonical SMILES: C12(C3(C4(C5(C3(C(C1(C5(C2(C4(Cl)Cl)Cl)Cl)Cl)(Cl)Cl)Cl)Cl)Cl)Cl)Cl
Structural Properties:
Molecular Formula: C10Cl12
Molecular Weight: 545.543
Pharmacophore Features:
Number of bond donors: 0
Number of bond acceptors: 0
Number of atoms different from hydrogen: 22
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Chen TT, Reid PC, van Beneden R, Sonstegard RA. 1986. Effect of Aroclor 1254 and mirex on estradiol-induced vitellogenin production in juvenile rainbow trout (Salmo gairdneri). Canadian Journal of Fisheries & Aquatic Sciences 43(1):169-173.
Chu I, Villeneuve DC, Secours VE, Valli VE, Becking GC. 1981. Effects of photomirex and mirex on reproduction in the rat. Toxicol Appl Pharmacol 60(3):549-556.
Jovanovich L, Levin S, Khan MAQ. 1987. Significance of mirex-caused hypoglycemia and hyperlipidemia in rats. J Biochem Toxicol 2:203-213.
Rogers JM, Morelli L, Grabowski CT. 1984. Plasma glucose and protein concentrations in rat fetuses and neonates exposed to cataractogenic doses of mirex. Environ Res 34(1):155-161.Yarbrough JD, Chambers JE, Grimley JM, Alley EG, Fang MM, Morrow JT, Ward BC, Conroy JD. 1981 Mar 30. Comparative study of 8-monohydromirex and mirex toxicity in male rats. Toxicol Appl Pharmacol 58(1):105-117.
External Links

2D-structure
3D-structure