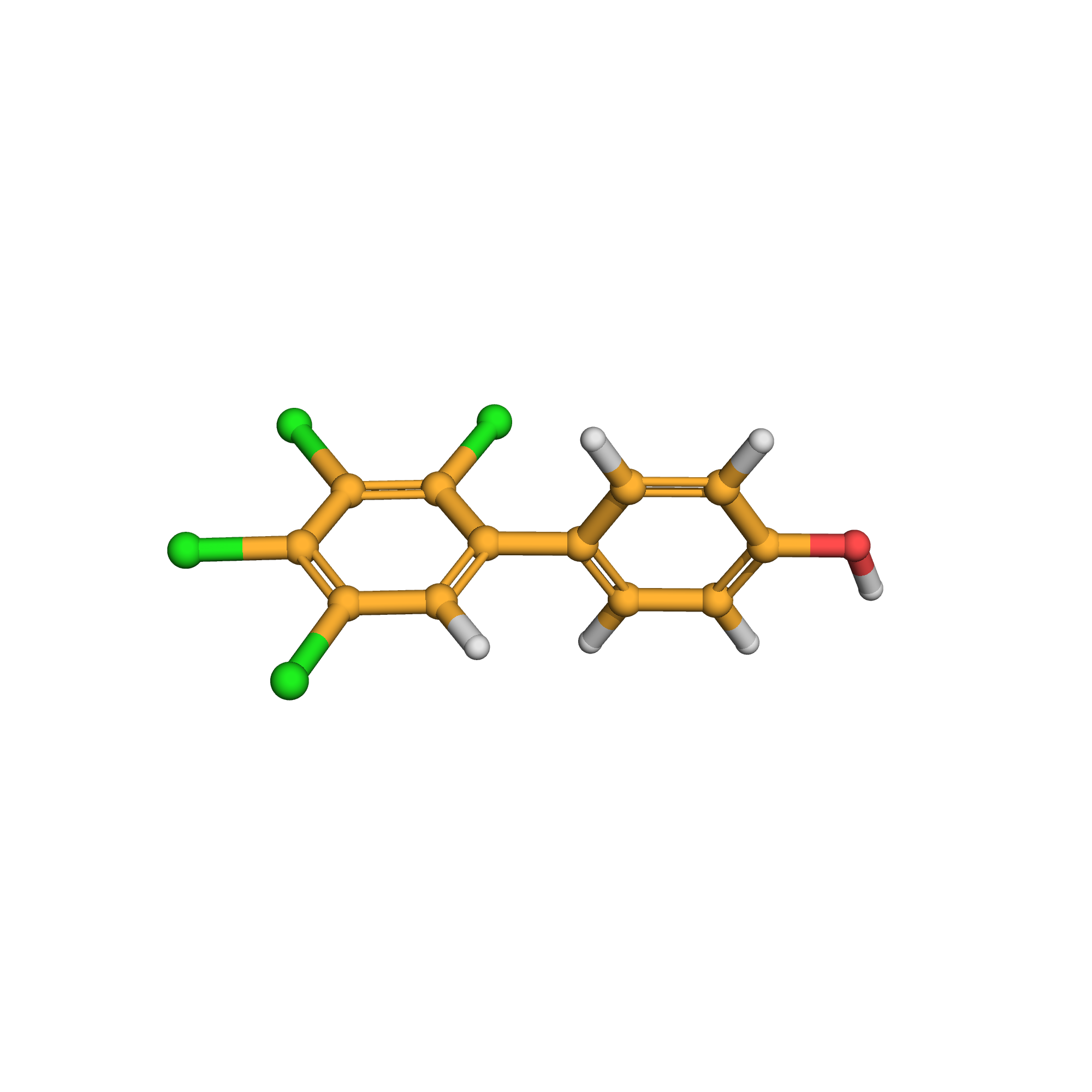
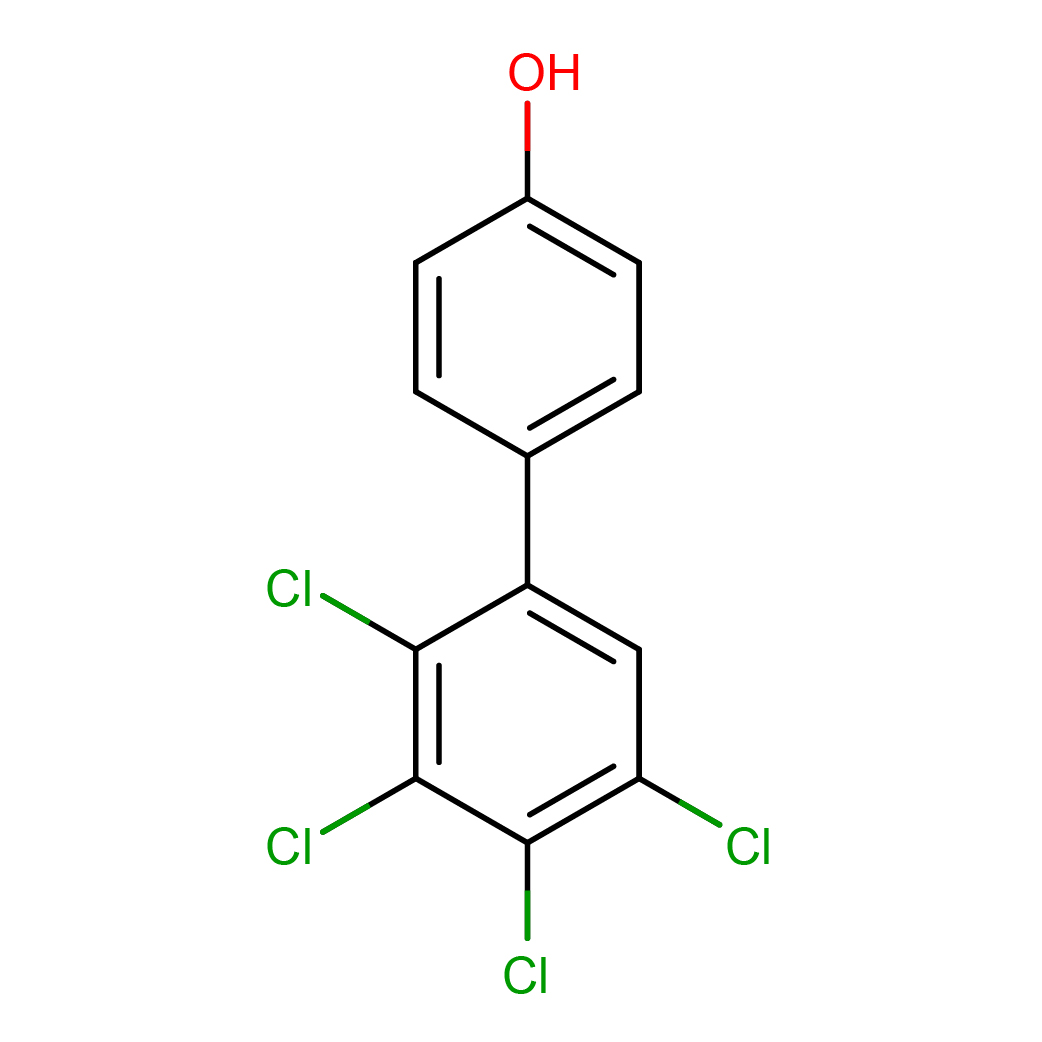
4'-hydroxy-2,3,4,5-tetrachlorobiphenyl
Synonyms: "4-hydroxy-2',3',4'-5'-tetrachlorobiphenyl", "2,3,4,5-tetrachloro-4'-biphenylol", "2',3',4',5'-tetrachloro-4-biphenylol", "OH-PCB-61"
Source: 4'-hydroxy-2,3,4,5-tetrachlorobiphenyl belongs to the hydroxylated metabolites of polychlorinated biphenyls (OH-PCBs).
Identifiers:
IUPAC Name: 4-(2,3,4,5-tetrachlorophenyl)phenol
CAS Number: 67651-34-7
PubChem ID: 105101
InChiKey: RESKVBRHSNTJNG-UHFFFAOYSA-N
Canonical SMILES: C1=CC(=CC=C1C2=CC(=C(C(=C2Cl)Cl)Cl)Cl)O
Structural Properties:
Molecular Formula: C12H6Cl4O
Molecular Weight: 307.987
Pharmacophore Features:
Number of bond donors: 1
Number of bond acceptors: 1
Number of atoms different from hydrogen: 17
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Cheek AO, Kow K, Chen J, McLachlan JA. 1999. Potential mechanisms of thyroid disruption in humans: Interaction of organochlorine compounds with thyroid receptor, transthyretin, and thyroid-binding globulin. Environ Health Perspect 107(4):273-278.
Jin L, Tran DQ, Ide CF, McLachlan JA, Arnold SF. 1997. Several synthetic chemicals inhibit progesterone receptor-mediated transactivation in yeast. Biochemical & Biophysical Research Communications 233(1):139-146.
Kramer VJ, Giesy JP. 1999. Specific binding of hydroxylated polychlorinated biphenyl metabolites and other substances to bovine calf uterine estrogen receptor: structure-binding relationships. Sci Total Environ 233(1-3):141-161.
Soto AM, Sonnenschein C, Chung KL, Fernandez MF, Olea N, Serrano FO. 1995. The E-SCREEN assay as a tool to identify estrogens: An update on estrogenic environmental pollutants. Environ Health Perspect 103 (Suppl. 7):113-122.
External Links


2D-structure
3D-structure