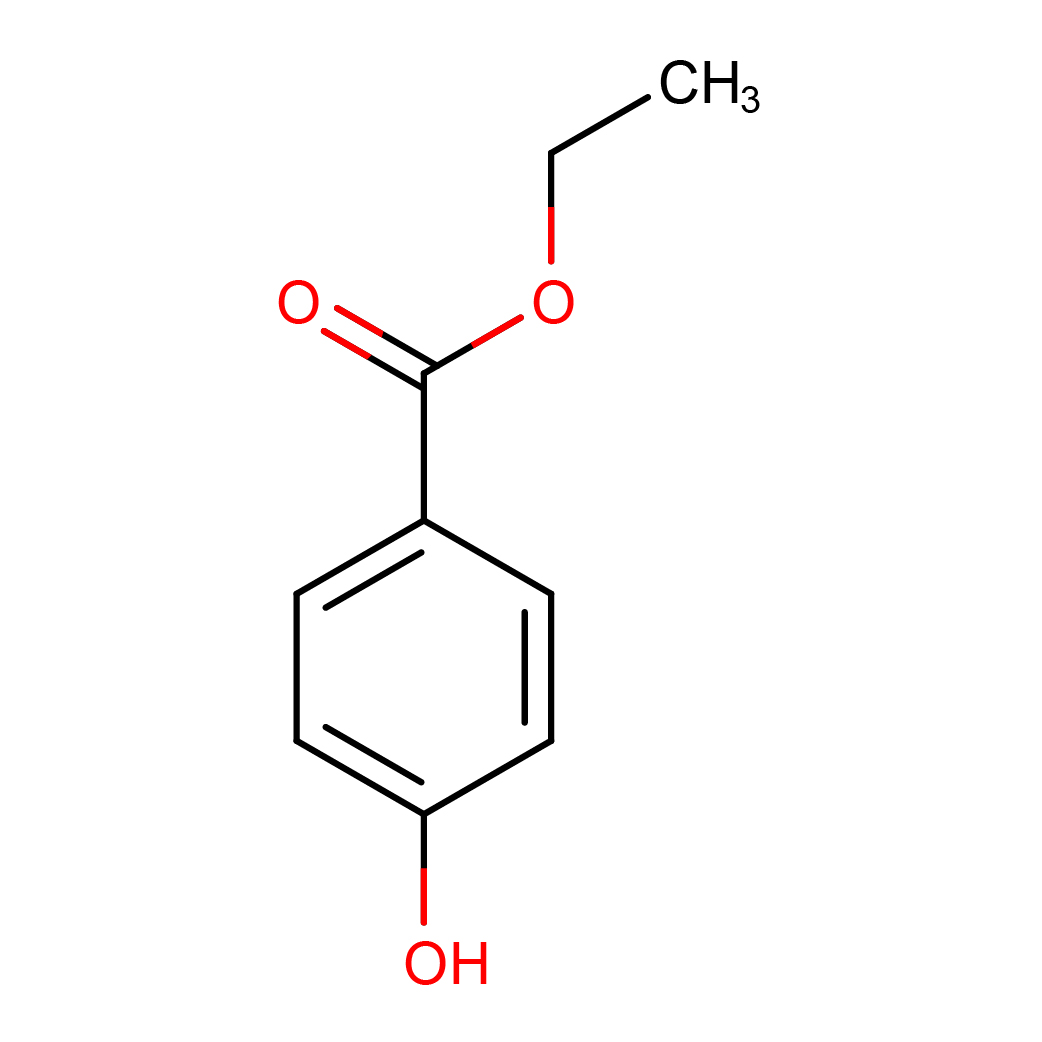
ethyl paraben
Synonyms: "ethyl-p-hydroxybenzoate", "ethyl 4-hydroxybenzoate", "ethyl p-hydroxybenzoate", "easeptol", "mycocten", "ethyl parasept", "ethyl butex"
Source: ethyl paraben is used as a preservative.
Identifiers:
IUPAC Name: ethyl 4-hydroxybenzoate
CAS Number: 120-47-8
PubChem ID: 8434
InChiKey: NUVBSKCKDOMJSU-UHFFFAOYSA-N
Canonical SMILES: CCOC(=O)C1=CC=C(C=C1)O
Structural Properties:
Molecular Formula: C9H10O3
Molecular Weight: 166.174
Pharmacophore Features:
Number of bond donors: 1
Number of bond acceptors: 2
Number of atoms different from hydrogen: 12
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Byford JR, Shaw LE, Drew MG, Pope GS, Sauer MJ, Darbre PD. 2002. Oestrogenic activity of parabens in MCF7 human breast cancer cells. Journal of Steroid Biochemistry & Molecular Biology 80(1):49-60.
Gomez E, Pillon A, Fenet H, Rosain D, Duchesne MJ, Nicolas JC, Balaguer P, Casellas C. 2005. Estrogenic activity of cosmetic components in reporter cell lines: parabens, UV screens, and musks. J Toxicol Environ Health A 68(4):239-251.
Lemini C, Herná,ndez A, Jaimez R, Franco Y, Avila ME, Castell A. 2004. Morphometric analysis of mice uteri treated with the preservatives methyl, ethyl, propyl, and butylparaben. Toxicol Ind Health 20(6-10):123-132.
Song BL, Li HY, Peng DR. 1989. In vitro spermicidal activity of parabens against human spermatozoa. Contraception 39(3):331-335.
Taxvig C, Vinggaard AM, Hass U, Axelstad M, Boberg J, Hansen PR, Frederiksen H, Nellemann C. 2008. Do parabens have the ability to interfere with steroidogenesis? Toxicol Sci 106(1):206-213.
External Links

2D-structure
3D-structure