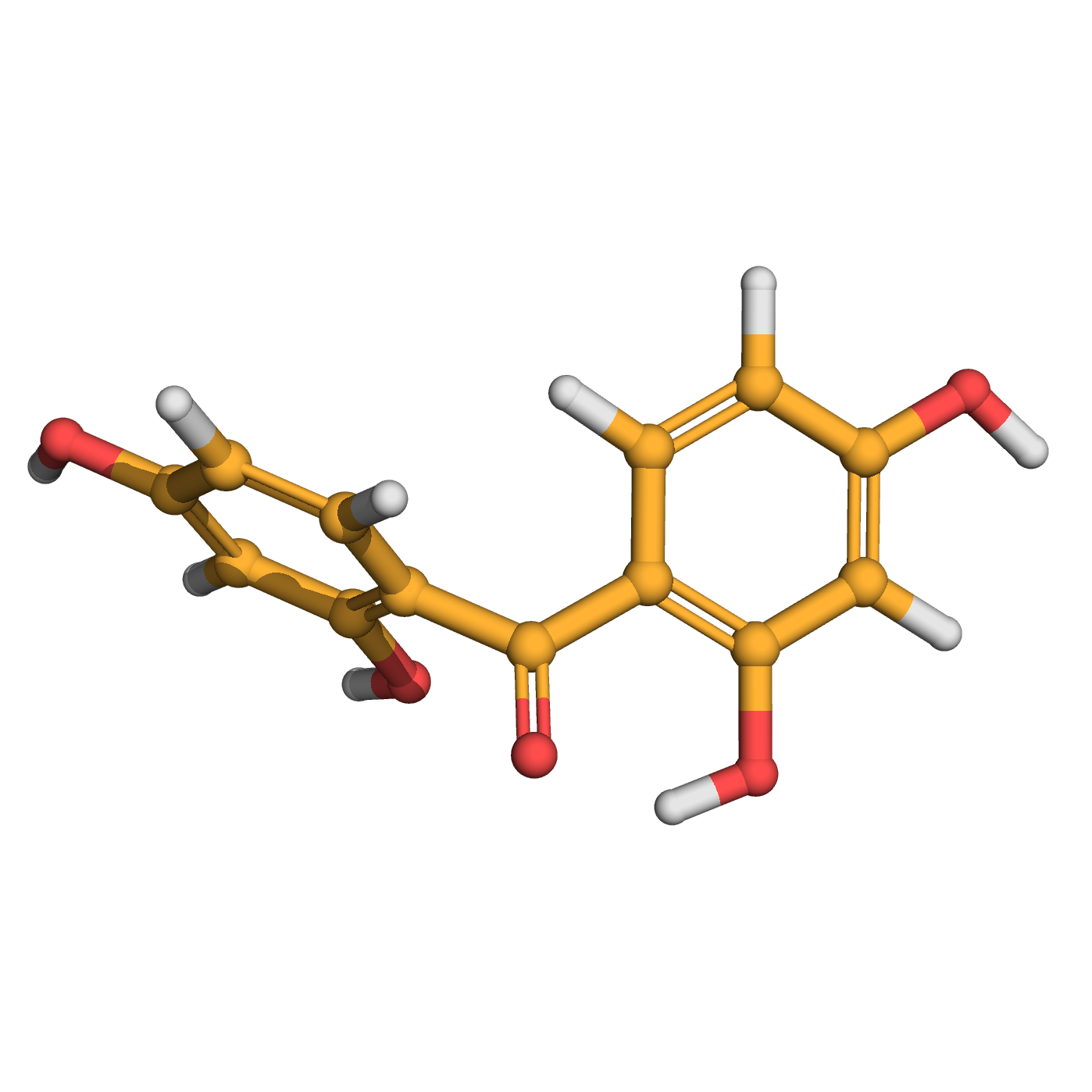
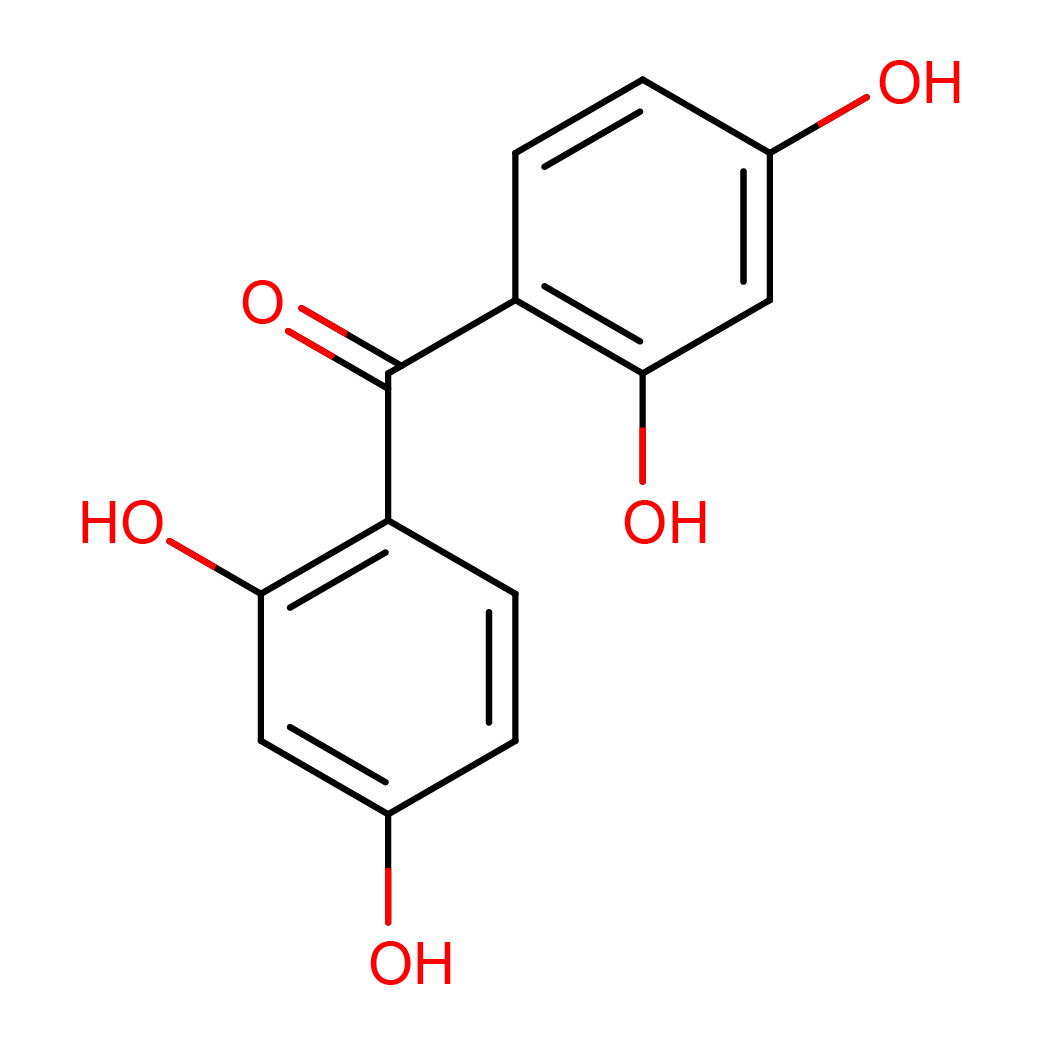
2,2',4,4'-tetrahydroxybenzophenone
Synonyms: "benzophenone-2", "bis(2,4-dihydroxyphenyl)methanone", "uvinol D-50"
Source: 2,2',4,4'-tetrahydroxybenzophenone is an effective ultraviolet light absorber (UV absorber) which acts as a stabilizer for protecting plastics, coatings and cosmetics from ultraviolet light (UV) radiation damage found in sunlight.
Identifiers:
IUPAC Name: bis(2,4-dihydroxyphenyl)methanone
CAS Number: 131-55-5
PubChem ID: 8571
InChiKey: WXNRYSGJLQFHBR-UHFFFAOYSA-N
Canonical SMILES: C1=CC(=C(C=C1O)O)C(=O)C2=C(C=C(C=C2)O)O
Structural Properties:
Molecular Formula: C13H10O5
Molecular Weight: 246.216
Pharmacophore Features:
Number of bond donors: 4
Number of bond acceptors: 5
Number of atoms different from hydrogen: 18
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Molina-Molina JM, Escande A, Pillon A, Gomez E, Pakdel F, Cavailles V, Olea N, Ait-Aissa S, Balaguer P. 2008. Profiling of benzophenone derivatives using fish and human estrogen receptor-specific in vitro bioassays. Toxicol Appl Pharmacol 232(3):384-395.
Schmutzler C, Bacinski A, Gotthardt I, Huhne K, Ambrugger P, Klammer H, Schlecht C, Hoang-Vu C, Gruters A, Wuttke W, Jarry H, Kohrle J. 2007. The ultraviolet filter benzophenone 2 interferes with the thyroid hormone axis in rats and is a potent in vitro inhibitor of human recombinant thyroid peroxidase. Endocrinology 148(6):2835-2844.
Schultz TW, Seward JR, Sinks GD. 2000. Estrogenicity of benzophenones evaluated with a recombinant yeast assay: comparison of experimental and rules-based predicted activity. Environ Toxicol Chem 19(2):301-304.
Seidlova-Wuttke D, Jarry H, Christoffel J, Rimoldi G, Wuttke W. 2005. Effects of bisphenol-A (BPA), dibutylphtalate (DBP), benzophenone-2 (BP2), procymidone (Proc), and linurone (Lin) on fat tissue, a variety of hormones and metabolic parameters: A 3 months comparison with effects of estradiol (E2) in ovariectomized (ovx) rats. Toxicology 213(1-2):13-24.Suzuki T, Kitamura S, Khota R, Sugihara K, Fujimoto N, Ohta S. 2005. Estrogenic and antiandrogenic activities of 17 benzophenone derivatives used as UV stabilizers and sunscreens. Toxicol Appl Pharmacol 203(1):9-17.
External Links


2D-structure
3D-structure