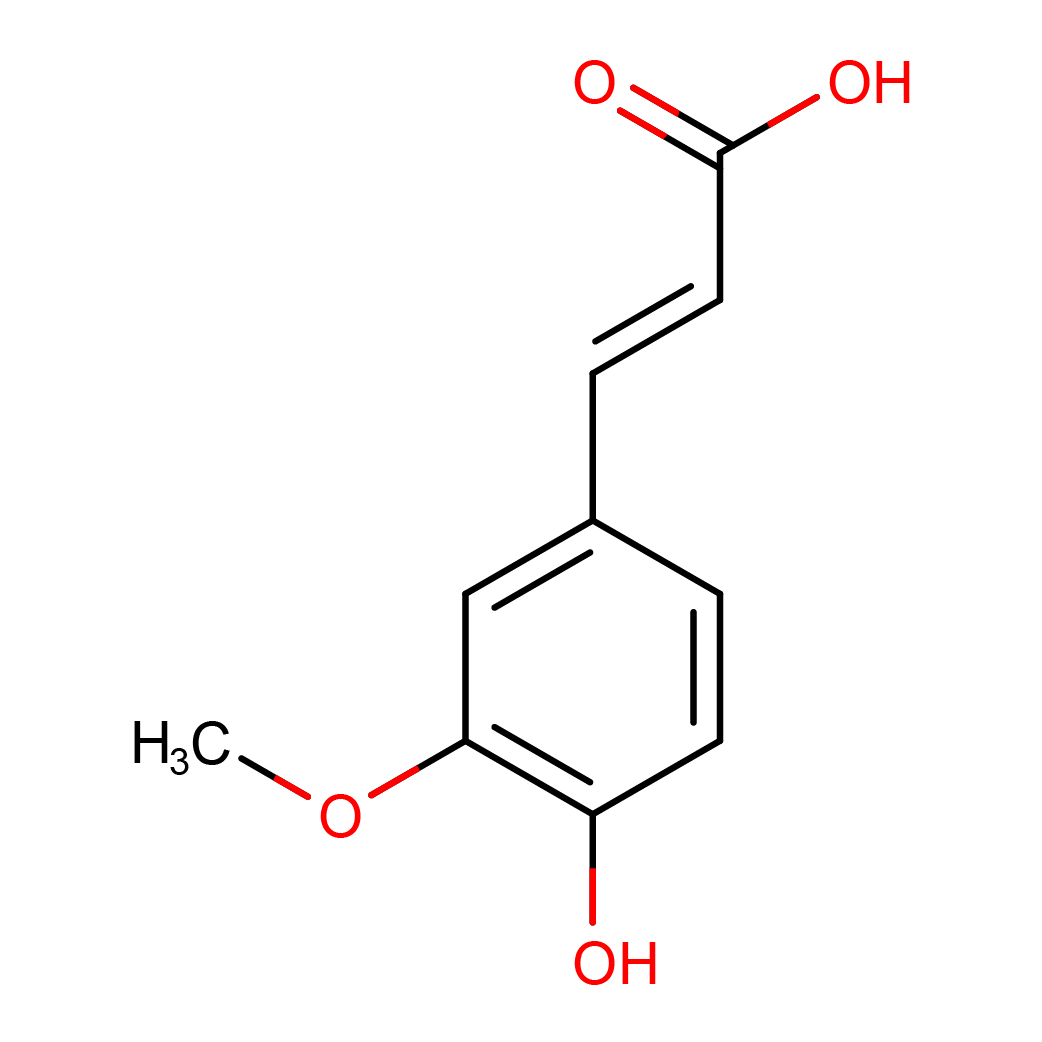
ferulic acid
Synonyms: "trans-ferulic acid", "4-hydroxy-3-methoxycinnamic acid", "ferulate", "(E)-ferulic acid", "coniferic acid", "trans-4-hydroxy-3-methoxycinnamic acid"
Source: ferulic acid is used in a wide range of cosmetics, such as skin lighteners, sunscreens, anti-aging creams and moisturizers.
Identifiers:
IUPAC Name: (E)-3-(4-hydroxy-3-methoxyphenyl)prop-2-enoic acid
CAS Number: 1135-24-6
PubChem ID: 445858
InChiKey: KSEBMYQBYZTDHS-HWKANZROSA-N
Canonical SMILES: COC1=C(C=CC(=C1)C=CC(=O)O)O
Structural Properties:
Molecular Formula: C10H10O4
Molecular Weight: 194.184
Pharmacophore Features:
Number of bond donors: 2
Number of bond acceptors: 4
Number of atoms different from hydrogen: 14
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Rosenberg RS, Grass L, Jenkins DJ, Kendall CWC, Diamandis EP. 1998. Modulation of androgen and progesterone receptors by phytochemicals in breast cancer cell lines. Biochemical & Biophysical Research Communications 248(3):935-939.
External Links

2D-structure
3D-structure