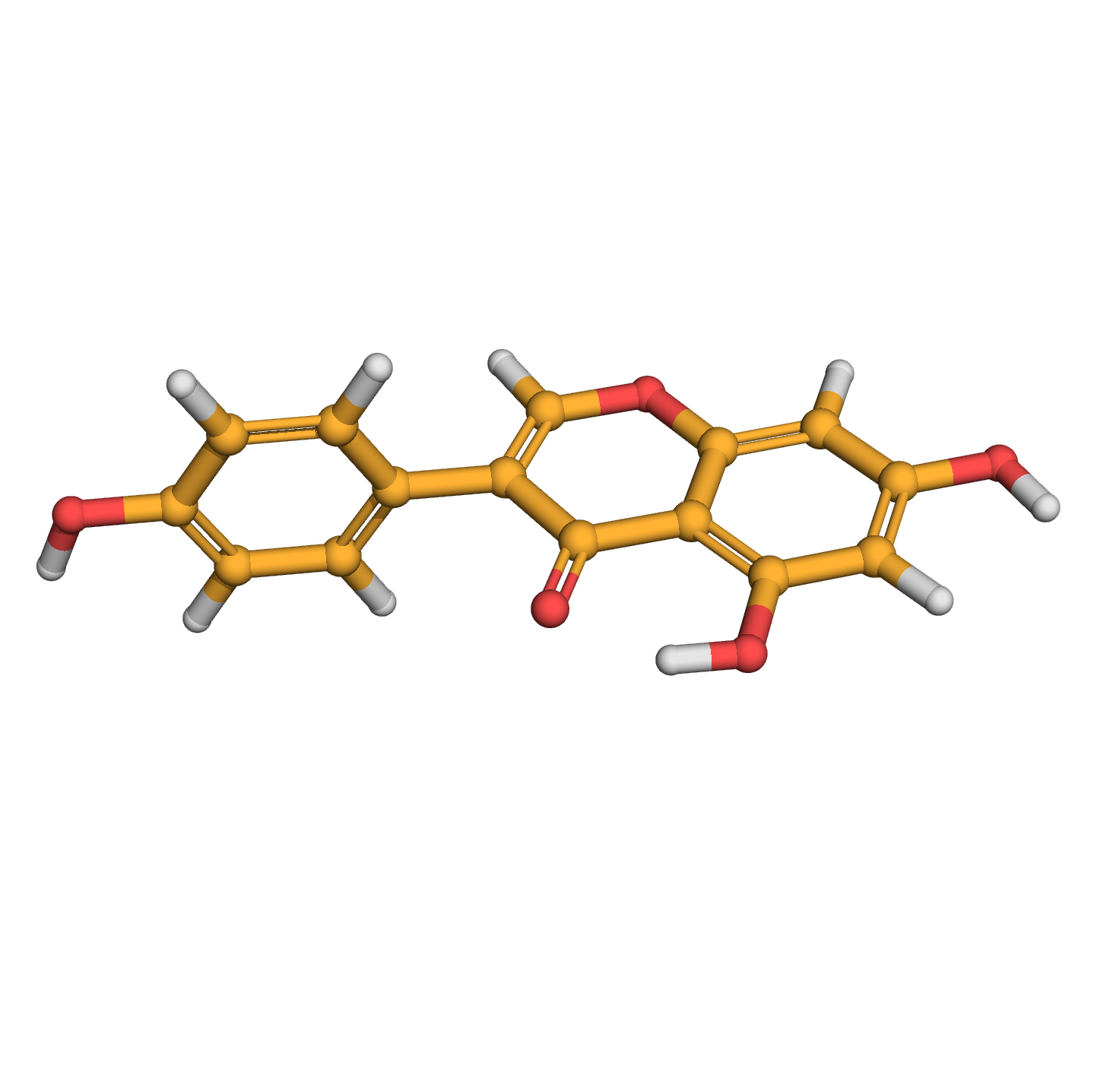
genistein
Synonyms: "prunetol", "genisteol", "genisterin", "sophoricol", "4',5,7-trihydroxyisoflavone", "5,7,4'-trihydroxyisoflavone", "differenol A", "genestein"
Source: genistein is a prominent isoflavonoid found in soy products .
Identifiers:
IUPAC Name: 5,7-dihydroxy-3-(4-hydroxyphenyl)chromen-4-one
CAS Number: 446-72-0
PubChem ID: 5280961
InChiKey: TZBJGXHYKVUXJN-UHFFFAOYSA-N
Canonical SMILES: C1=CC(=CC=C1C2=COC3=CC(=CC(=C3C2=O)O)O)O
Structural Properties:
Molecular Formula: C15H10O5
Molecular Weight: 270.237
Pharmacophore Features:
Number of bond donors: 3
Number of bond acceptors: 5
Number of atoms different from hydrogen: 20
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Davis JN, Kucuk O, Sarkar FH. 2002. Expression of prostate-specific antigen is transcriptionally regulated by genistein in prostate cancer cells. Mol Carcinog 34(2):91-101.
Nishihara T, Nishikawa J, Kanayama T, Dakeyama F, Saito K, Imagawa M, Takatori S, Kitagawa Y, Hori S, Utsumi H. 2000. Estrogenic activities of 517 chemicals by yeast two-hybrid assay. Journal of Health Science 46(4):282-298.
Ohlsson A, Ulleras E, Cedergreen N, Oskarsson A. 2010. Mixture effects of dietary flavonoids on steroid hormone synthesis in the human adrenocortical H295R cell line. Food Chem Toxicol 48(11):3194-3200.
Sanderson JT, Boerma J, Lansbergen GW, van den Berg M. 2002. Induction and inhibition of aromatase (CYP19) activity by various classes of pesticides in H295R human adrenocortical carcinoma cells. Toxicol Appl Pharmacol 182(1):44-54.
Scippo ML, Argiris C, Van De Weerdt C, Muller M, Willemsen P, Martial J, Maghuin-Rogister G. 2004. Recombinant human estrogen, androgen and progesterone receptors for detection of potential endocrine disruptors. Analytical & Bioanalytical Chemistry 378(3)
External Links


2D-structure
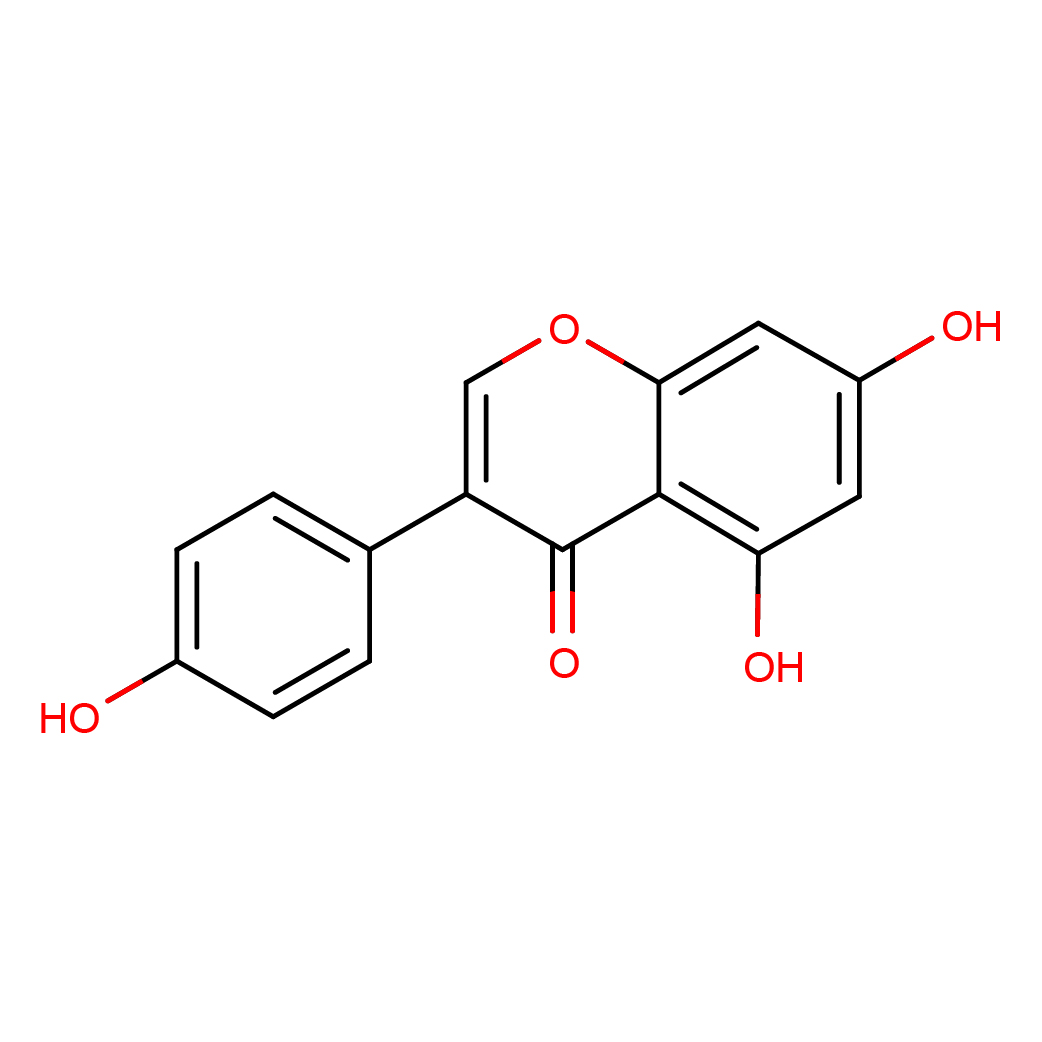
3D-structure