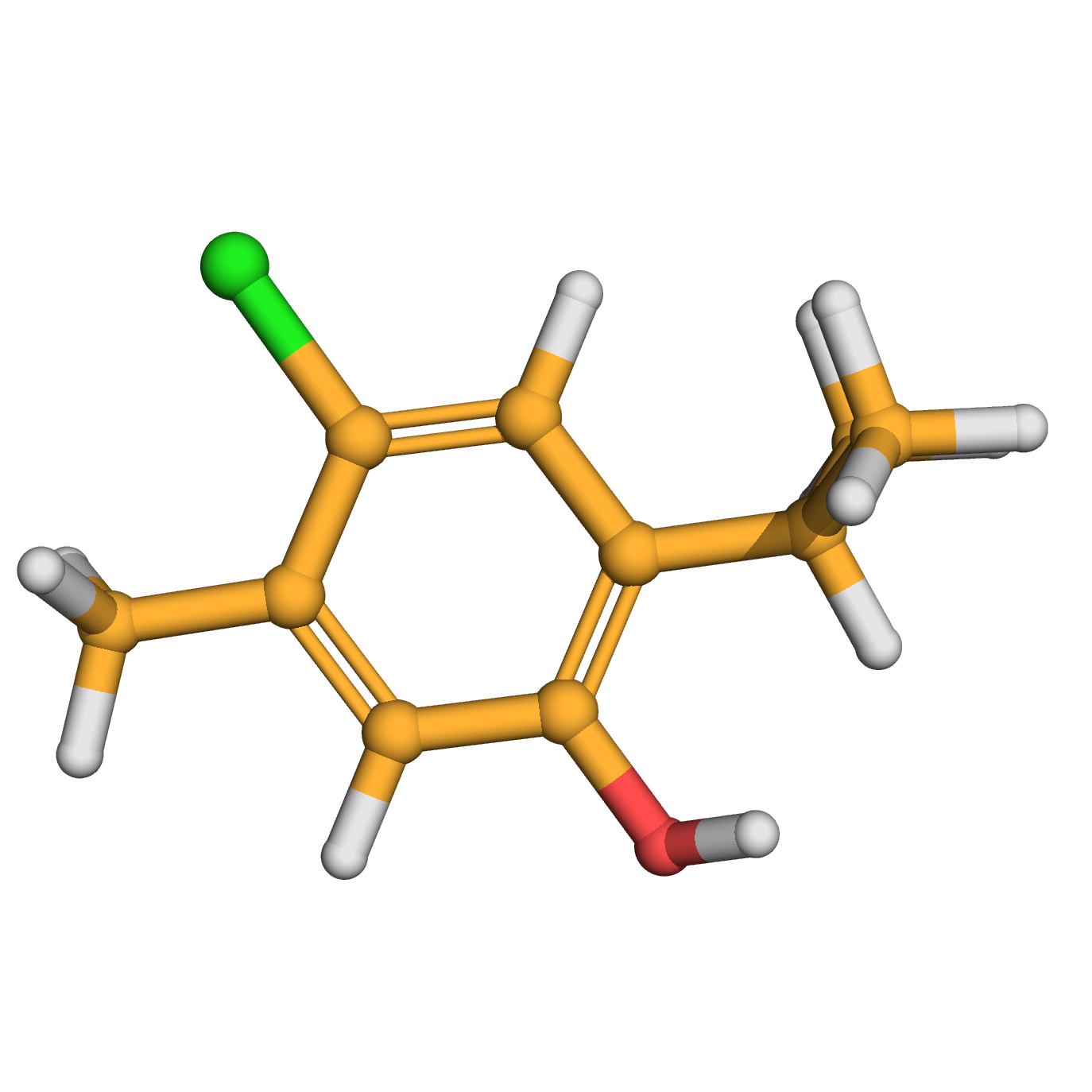
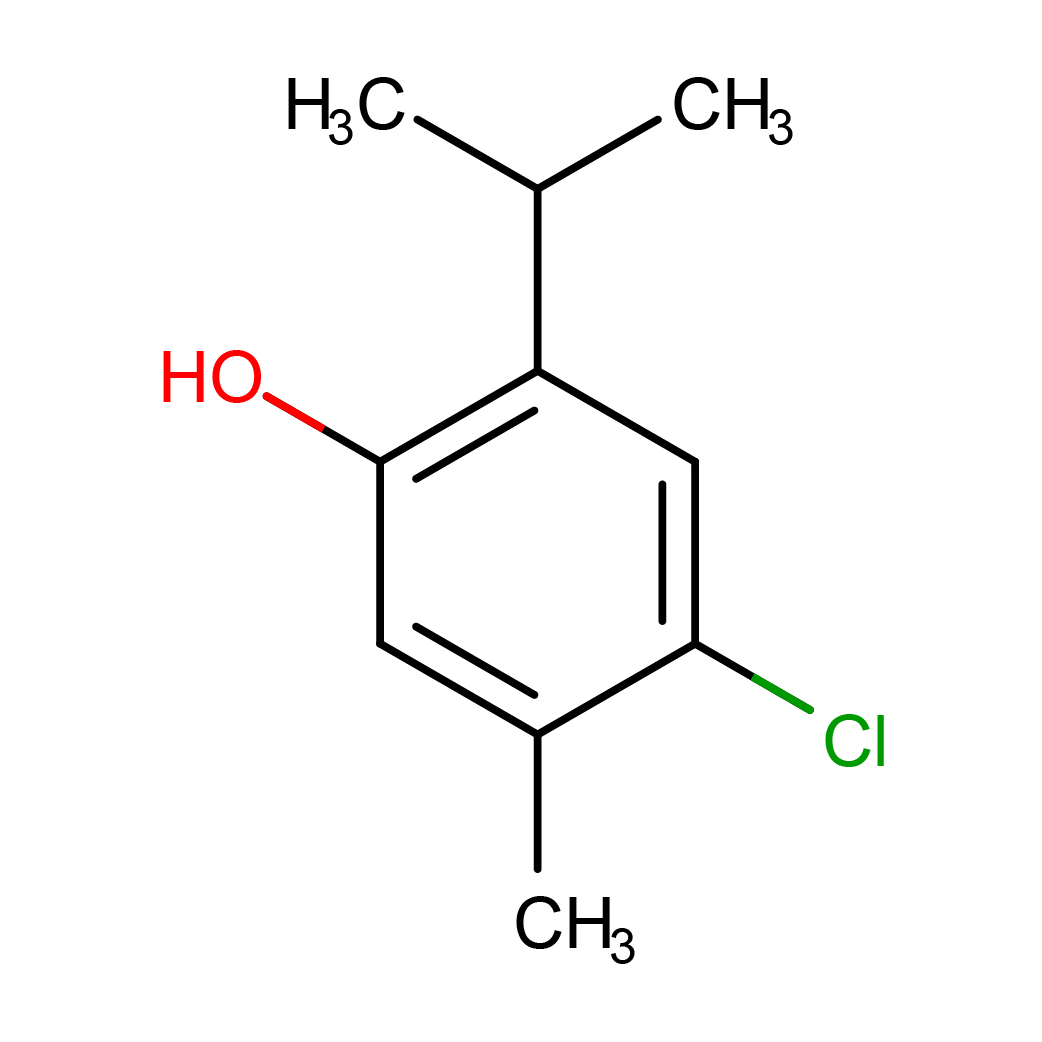
chlorothymol
Synonyms: "6-chlorothymol", "4-chloro-2-isopropyl-5-methylphenol", "chlorthymol", "4-chlorothymol", "6-chorothymol"
Source: chlorothymol is a chlorinated phenolic antiseptic used as an ingredient of preparations for hand and skin disinfection and topical treatment of fungal infections.
Identifiers:
IUPAC Name: 4-chloro-5-methyl-2-propan-2-ylphenol
CAS Number: 89-68-9
PubChem ID: 6982
InChiKey: KFZXVMNBUMVKLN-UHFFFAOYSA-N
Canonical SMILES: CC1=CC(=C(C=C1Cl)C(C)C)O
Structural Properties:
Molecular Formula: C10H13ClO
Molecular Weight: 184.663
Pharmacophore Features:
Number of bond donors: 1
Number of bond acceptors: 1
Number of atoms different from hydrogen: 12
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Kramer VJ, Giesy JP. 1999. Specific binding of hydroxylated polychlorinated biphenyl metabolites and other substances to bovine calf uterine estrogen receptor: structure-binding relationships. Sci Total Environ 233(1-3):141-161.
Miller D, Wheals BB, Beresford N, Sumpter JP. 2001. Estrogenic activity of phenolic additives determined by an in vitro yeast bioassay. Environ Health Perspect 109(2):133-138.
External Links

2D-structure
3D-structure