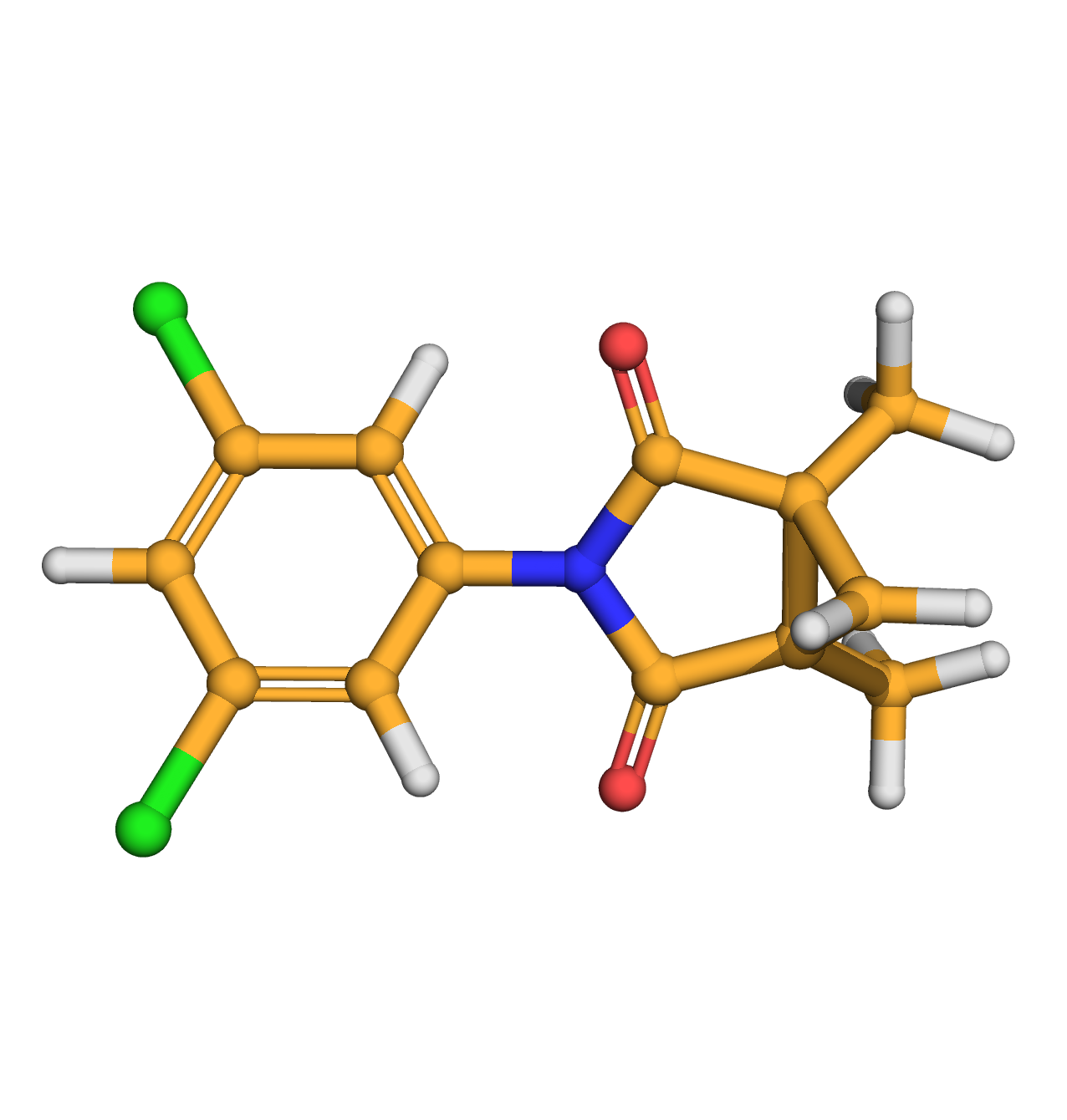
procymidone
Synonyms: "dicyclidine", "sumisclex", "sumilex", "3-(3,5-dichlorophenyl)-1,5-dimethyl-3-azabicyclo[3.1.0]hexane-2,4-dione"
Source: procymidone is a systemic fungicide used on lupins, grapes, stone fruit, strawberries and some vegetables.
Identifiers:
IUPAC Name: 3-(3,5-dichlorophenyl)-1,5-dimethyl-3-azabicyclo[3.1.0]hexane-2,4-dione
CAS Number: 32809-16-8
PubChem ID: 36242
InChiKey: QXJKBPAVAHBARF-UHFFFAOYSA-N
Canonical SMILES: CC12CC1(C(=O)N(C2=O)C3=CC(=CC(=C3)Cl)Cl)C
Structural Properties:
Molecular Formula: C13H11Cl2NO2
Molecular Weight: 284.138
Pharmacophore Features:
Number of bond donors: 0
Number of bond acceptors: 2
Number of atoms different from hydrogen: 18
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Kojima H, Katsura E, Takeuchi S, Niiyama K, Kobayashi K. 2004. Screening for estrogen and androgen receptor activities in 200 pesticides by in vitro reporter gene assays using Chinese hamster ovary cells. Environ Health Perspect 112(5):524-531.
Ostby J, Kelce WR, Lambright C, Wolf CJ, Mann P, Gray LE Jr. 1999. The fungicide procymidone alters sexual differentiation in the male rat by acting as an androgen-receptor antagonist in vivo and in vitro. Toxicol Ind Health 15(1-2):80-93.
External Links


2D-structure
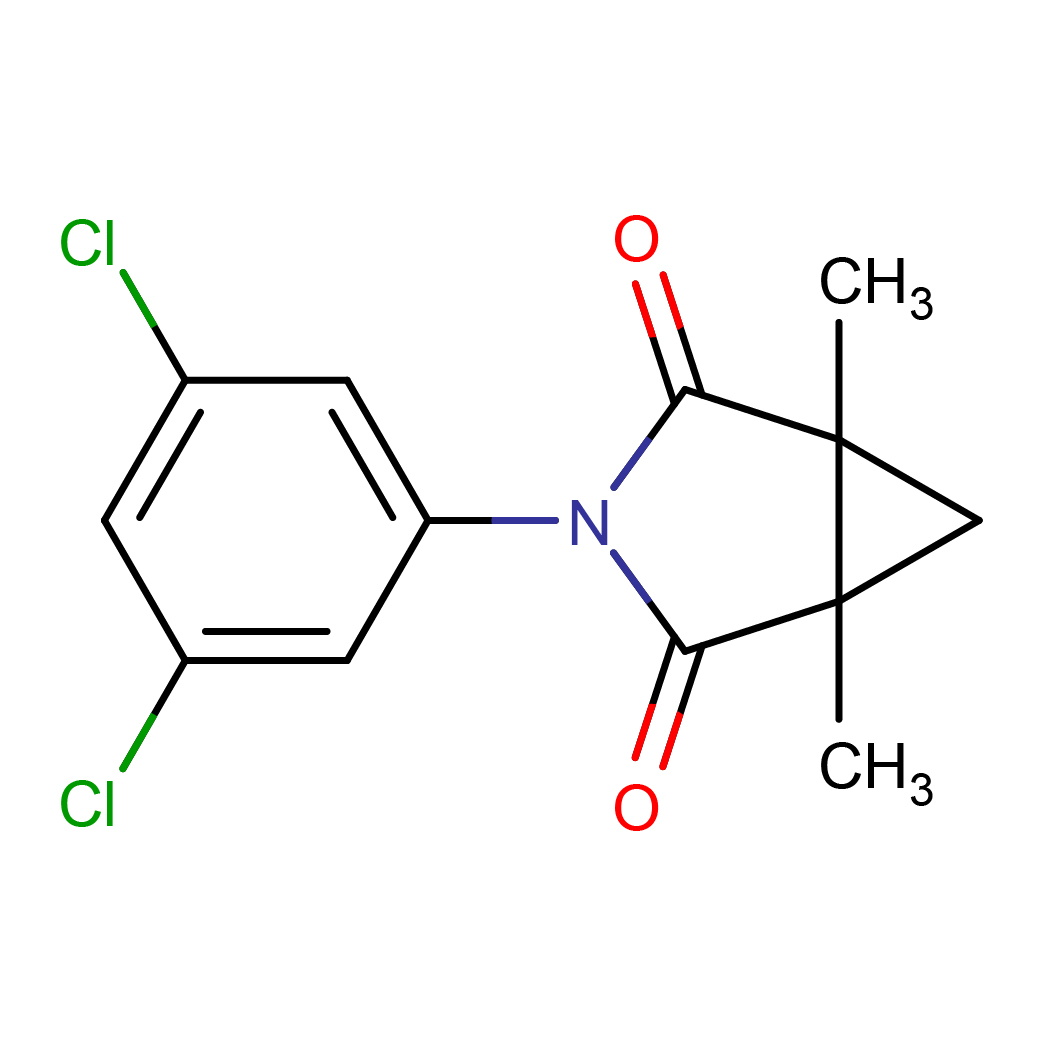
3D-structure