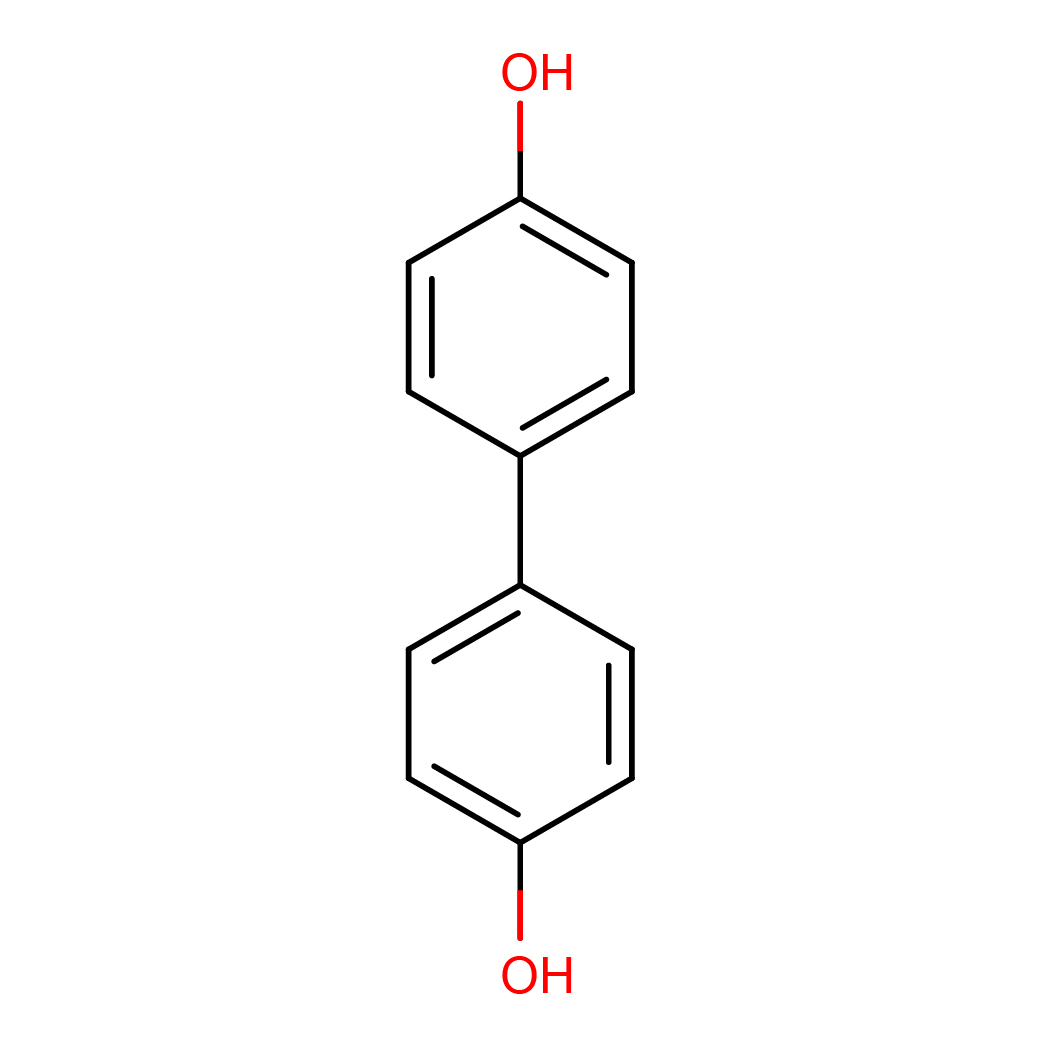
4,4'-biphenol
Synonyms: "4,4'-dihydroxybiphenyl ", "4,4'-biphenyldiol", "biphenyl-4,4'-diol", "p,p'-diphenol", "p,p'-biphenol", "4,4'-diphenol", "4,4'-dioxydiphenyl"
Source: 4,4'-biphenol is used to synthesize liquid crystal polymers (LCP).
Identifiers:
IUPAC Name: 4-(4-hydroxyphenyl)phenol
CAS Number: 92-88-6
PubChem ID: 7112
InChiKey: VCCBEIPGXKNHFW-UHFFFAOYSA-N
Canonical SMILES: C1=CC(=CC=C1C2=CC=C(C=C2)O)O
Structural Properties:
Molecular Formula: C12H10O2
Molecular Weight: 186.207
Pharmacophore Features:
Number of bond donors: 2
Number of bond acceptors: 2
Number of atoms different from hydrogen: 14
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Akahori Y, Nakai M, Yamasaki K, Takatsuki M, Shimohigashi Y, Ohtaki M. 2008. Relationship between the results of in vitro receptor binding assay to human estrogen receptor alpha and in vivo uterotrophic assay: Comparative study with 65 selected chemicals. Toxicol in Vitro 22(1):225-231.
Han D-H, Tachibana H, Yamada K. 2001. Inhibition of environmental estrogen-induced proliferation of human breast carcinoma MCF-7 cells by flavonoids. In Vitro Cellular & Developmental Biology-Animal 37(5):275-282.
Olsen CM, Meussen-Elholm ETM, Samuelsen M, Holme JA, Hongslo JK. 2003. Effects of the environmental oestrogens bisphenol A, tetrachlorobisphenol A, tetrabromobisphenol A, 4-hydroxybiphenyl and 4,4 '-dihydroxybiphenyl on oestrogen receptor binding, cell proliferation and regulation of oestrogen sensitive proteins in the human breast cancer cell line MCF-7. Pharmacology & Toxicology 92(4):180-188.
Paris F, Balaguer P, Terouanne B, Servant N, Lacoste C, Cravedi JP, Nicolas JC, Sultan C. 2002. Phenylphenols, biphenols, bisphenol-A and 4-tert-octylphenol exhibit alpha and beta estrogen activities and antiandrogen activity in reporter cell lines. Molecular & Cellular Endocrinology 193(1-2. Si):43-49.
Yamasaki K, Noda S, Imatanaka N, Yakabe Y. 2004. Comparative study of the uterotrophic potency of 14 chemicals in a uterotrophic assay and their receptor-binding affinity. Toxicol Lett 146(2):111-120.
External Links
2D-structure
3D-structure




