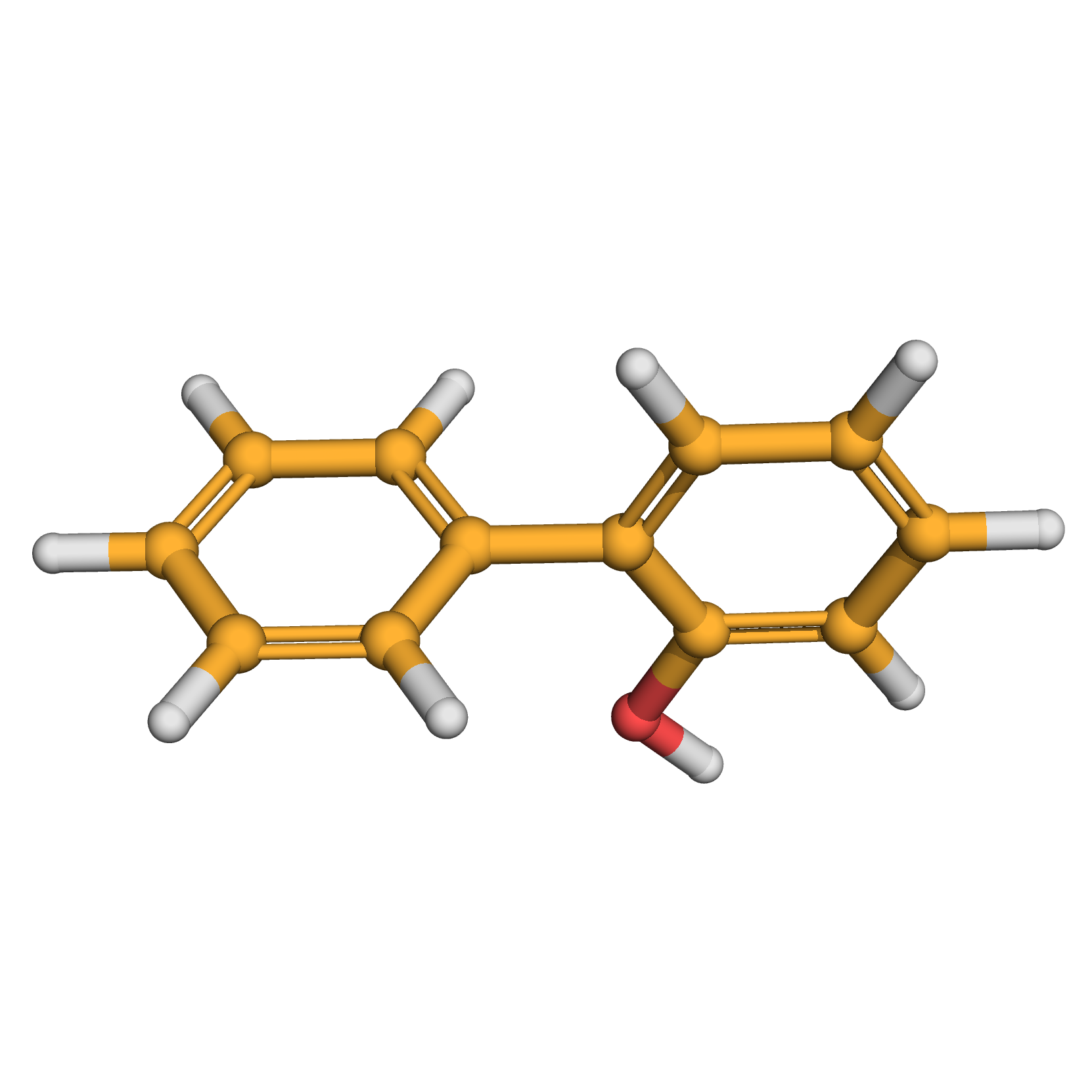
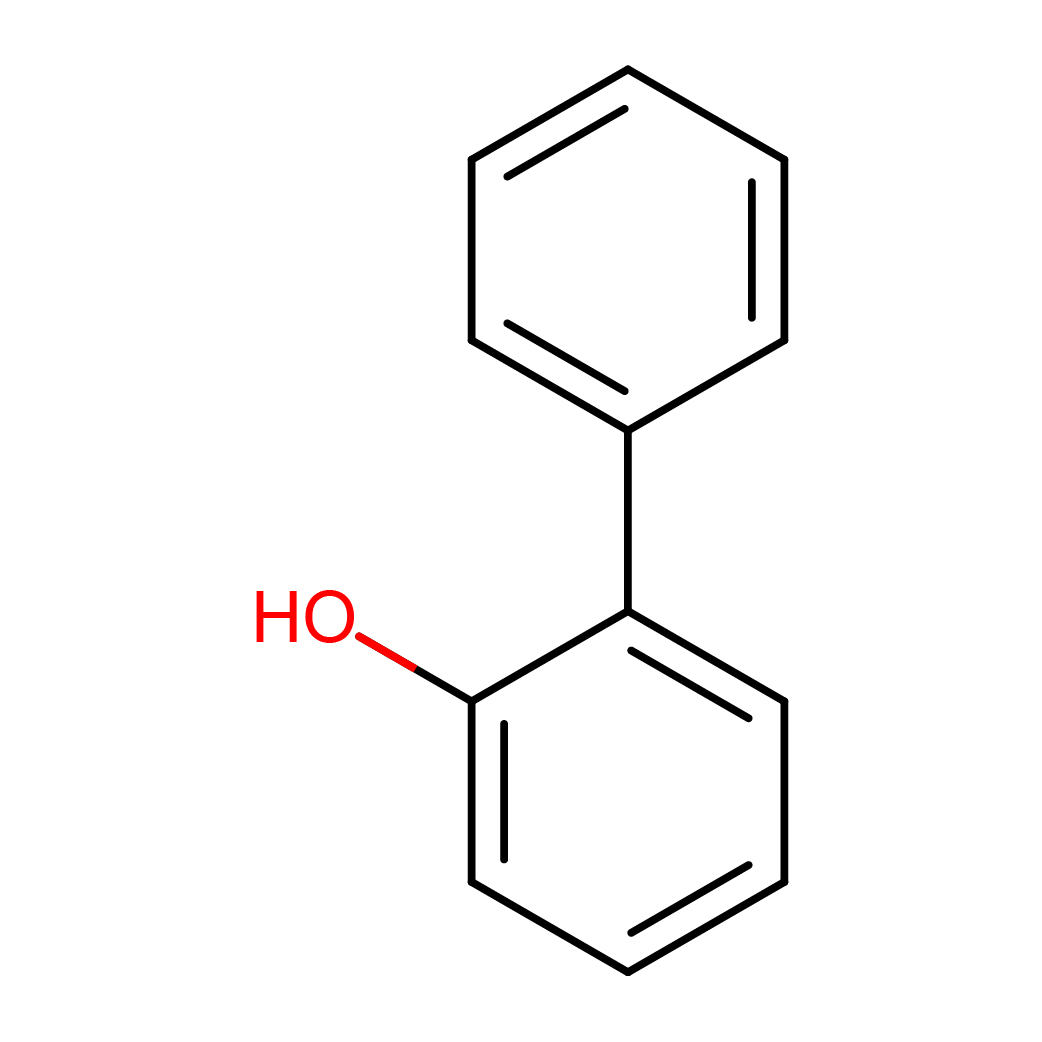
2-phenylphenol
Synonyms: "2-hydroxybiphenyl", "o-hydroxybiphenyl", "o-phenylphenol", "biphenyl-2-ol", "2-biphenylol", "2-hydroxydiphenyl", "biphenylol", "o-hydroxydiphenyl"
Source: 2-phenylphenol is an active ingredient of registered disinfecting products.
Identifiers:
IUPAC Name: 2-phenylphenol
CAS Number: 90-43-7
PubChem ID: 7017
InChiKey: LLEMOWNGBBNAJR-UHFFFAOYSA-N
Canonical SMILES: C1=CC=C(C=C1)C2=CC=CC=C2O
Structural Properties:
Molecular Formula: C12H10O
Molecular Weight: 170.207
Pharmacophore Features:
Number of bond donors: 1
Number of bond acceptors: 1
Number of atoms different from hydrogen: 13
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Cappelletti V, Saturno G, Miodini P, Korner W, Daidone MG. 2003. Selective modulation of ER-beta by estradiol and xenoestrogens in human breast cancer cell lines. Cellular & Molecular Life Sciences 60(3):567-576.
Dé,chaud H, Ravard C, Claustrat F, de la Perriè,re AB, Pugeat M. 1999. Xenoestrogen interaction with human sex hormone-binding globulin (hSHBG). Steroids 64(5):328-334.
Kojima H, Katsura E, Takeuchi S, Niiyama K, Kobayashi K. 2004. Screening for estrogen and androgen receptor activities in 200 pesticides by in vitro reporter gene assays using Chinese hamster ovary cells. Environ Health Perspect 112(5):524-531.
Okubo T, Kano I. 2003. [Studies on estrogenic activities of food additives with human breast cancer MCF-7 cells and mechanism of estrogenicity by BHA and OPP]. Yakugaku Zasshi 123(6):443-452.
Van den Berg KJ, van Raaij JAGM, Bragt PC, Notten WRF. 1991. Interactions of halogenated industrial chemicals with transthyretin and effects on thyroid hormone levels in vivo. Arch Toxicol 65(1):15-19.
External Links

2D-structure
3D-structure