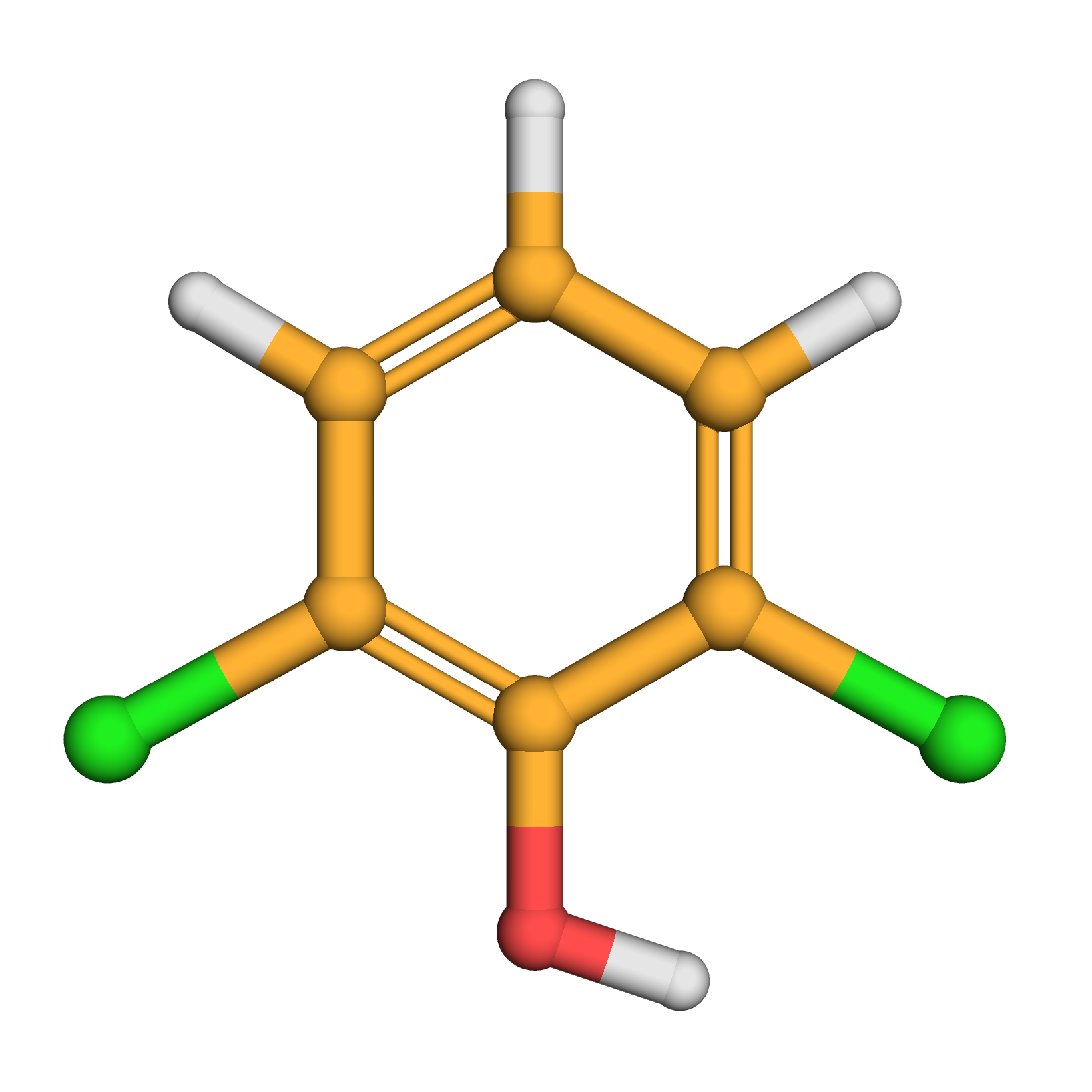
2,6-dichlorophenol
Synonyms: "2,6-dichloro-phenol", "2,6-DCP"
Source: 2,6-dichlorophenol is used as an intermediate in making insecticides, herbicides, preservatives, antiseptics, disinfectants and other organic compounds.
Identifiers:
IUPAC Name: 2,6-dichlorophenol
CAS Number: 87-65-0
PubChem ID: 6899
InChiKey: HOLHYSJJBXSLMV-UHFFFAOYSA-N
Canonical SMILES: C1=CC(=C(C(=C1)Cl)O)Cl
Structural Properties:
Molecular Formula: C6H4Cl2O
Molecular Weight: 163.001
Pharmacophore Features:
Number of bond donors: 1
Number of bond acceptors: 1
Number of atoms different from hydrogen: 9
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

den Besten C, Vet JJRM, Besselink HT, Kiel GS, Beems R, van Bladeren PJV. 1991. The liver, kidney, and thyroid toxicity of chlorinated benzenes. Toxicol Appl Pharmacol 111(1):69-81.
Van den Berg KJ, van Raaij JAGM, Bragt PC, Notten WRF. 1991. Interactions of halogenated industrial chemicals with transthyretin and effects on thyroid hormone levels in vivo. Arch Toxicol 65(1):15-19.
van den Berg KJ. 1990. Interaction of chlorinated phenols with thyroxine binding sites of human transthyretin, albumin and thyroid binding globulin. Chem Biol Interact 76(1):63-75.
External Links


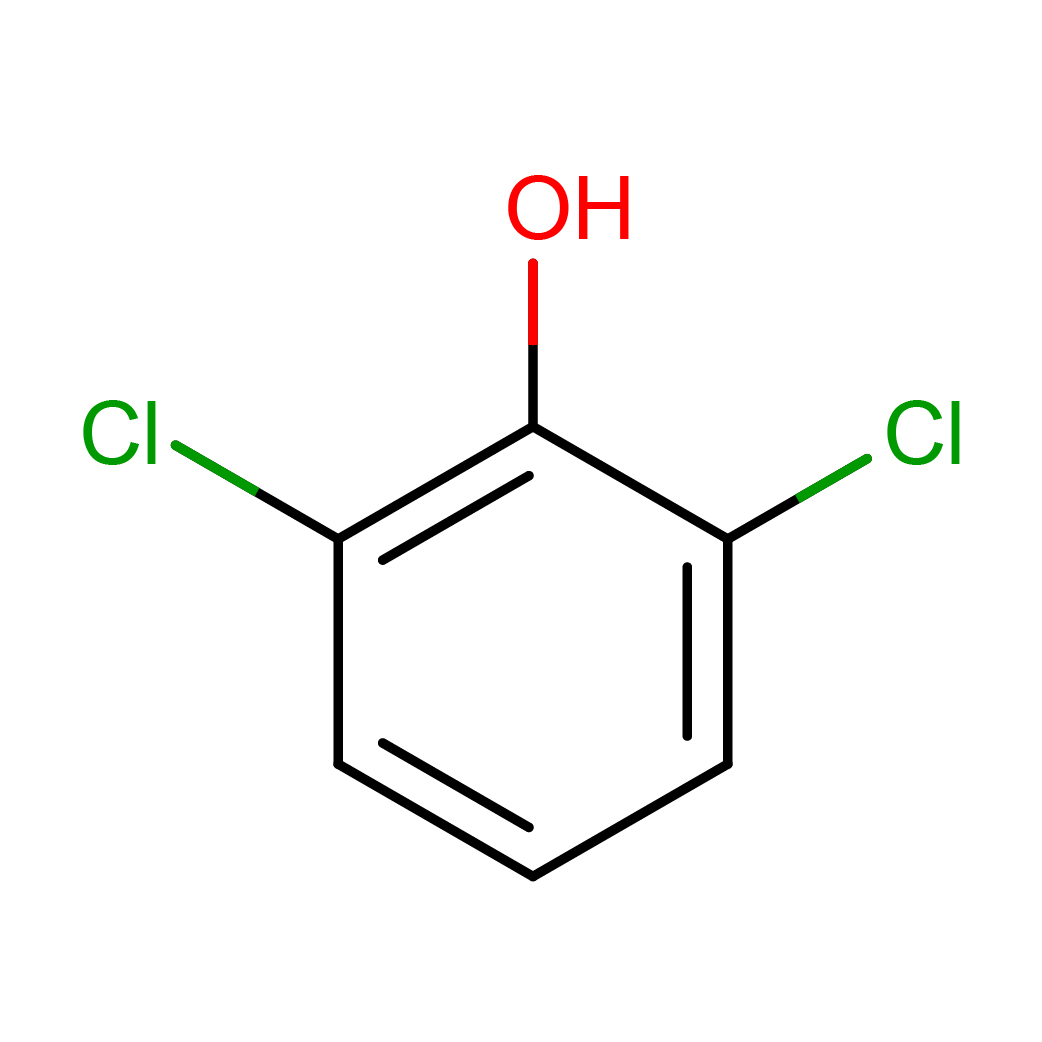
2D-structure
3D-structure