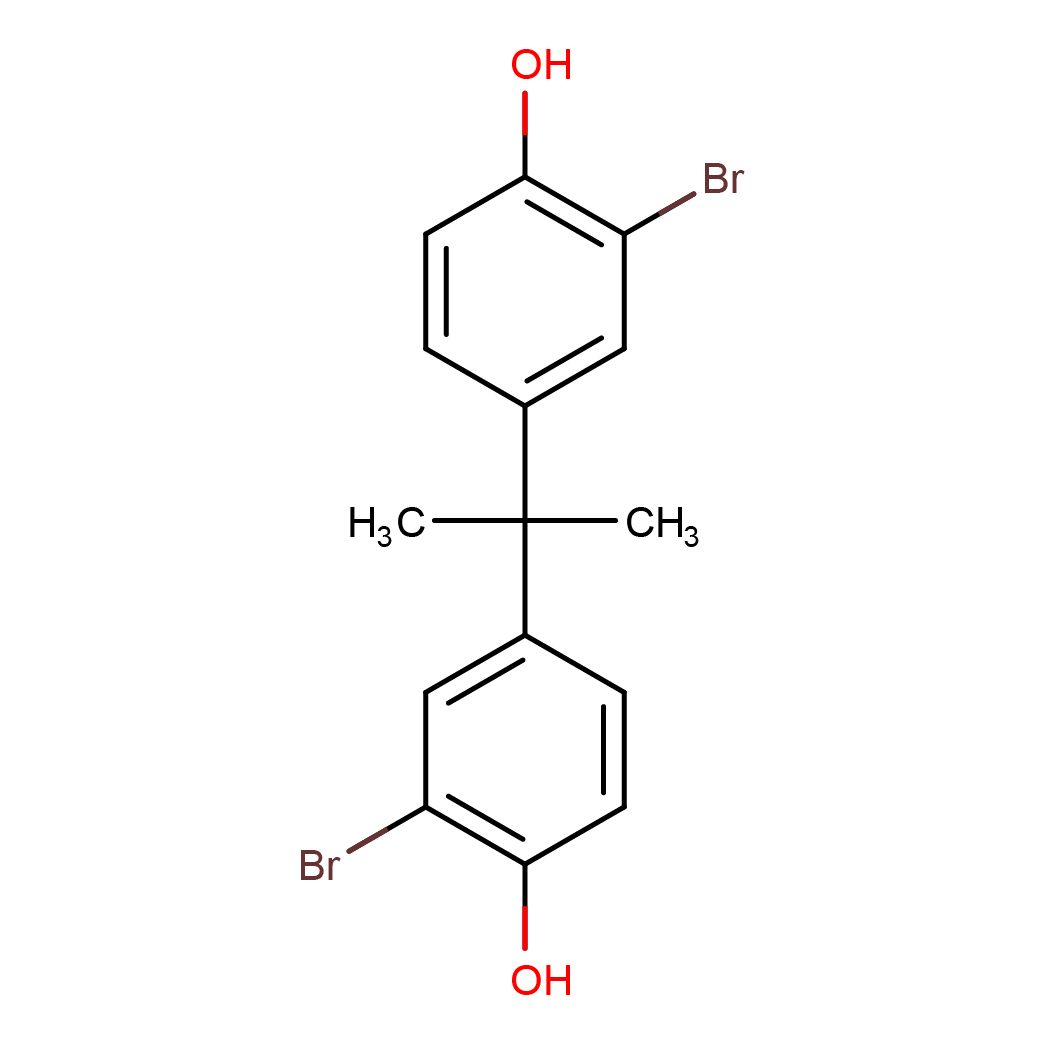
3,3'-dibromobisphenol A
Synonyms: "dibromobisphenol A", "2-bromo-4-[1-(3-bromo-4-hydroxyphenyl)-1-methylethyl]phenol", "4,4'-(propane-2,2-diyl)bis(2-bromophenol)"
Source: 3,3'-dibromobisphenol A is a derivative of tetrabromobisphenol A, which is one of the most commonly used brominated flame retardants worldwide.
Identifiers:
IUPAC Name: 2-bromo-4-[2-(3-bromo-4-hydroxyphenyl)propan-2-yl]phenol
CAS Number: 29426-78-6
PubChem ID: 656687
InChiKey: CKNCVRMXCLUOJI-UHFFFAOYSA-N
Canonical SMILES: CC(C)(C1=CC(=C(C=C1)O)Br)C2=CC(=C(C=C2)O)Br
Structural Properties:
Molecular Formula: C15H14Br2O2
Molecular Weight: 386.078
Pharmacophore Features:
Number of bond donors: 2
Number of bond acceptors: 2
Number of atoms different from hydrogen: 19
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Meerts IATM, Letcher RJ, Hoving S, Marsh G, Bergman Å,, Lemmen JG, van der Burg B, Brouwer A. 2001. In vitro estrogenicity of polybrominated diphenyl ethers, hydroxylated PBDEs, and polybrominated bisphenol A compounds. Environ Health Perspect 109(4):399-407, DOI: 10.2307/3454900.
Samuelsen M, Olsen C, Holme JA, Meussen-Elholm E, Bergmann A, Hongslo JK. 2001. Estrogen-like properties of brominated analogs of bisphenol A in the MCF-7 human breast cancer cell line. Cell Biology Toxicology 17(3):139-151.
External Links


2D-structure
3D-structure