luteolin
Synonyms: "luteolol", "3',4',5,7-tetrahydroxyflavone", "digitoflavone", "flacitran", "luteoline", "salifazide", "5,7,3',4'-tetrahydroxyflavone"
Source: luteolin is a flavonoid, known for its powerful antioxidant and anti-inflammatory properties.
Identifiers:
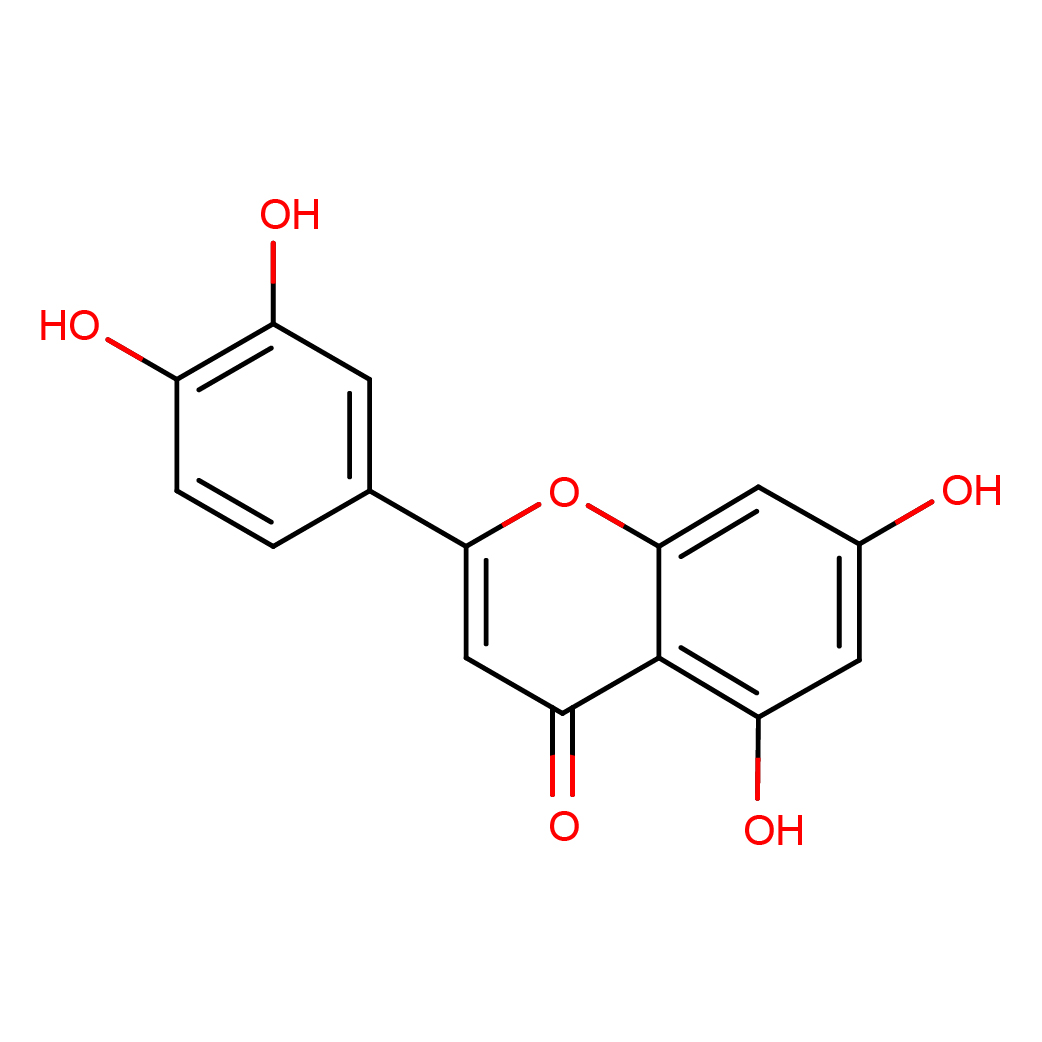
IUPAC Name: 2-(3,4-dihydroxyphenyl)-5,7-dihydroxychromen-4-one
CAS Number: 491-70-3
PubChem ID: 5280445
InChiKey: IQPNAANSBPBGFQ-UHFFFAOYSA-N
Canonical SMILES: C1=CC(=C(C=C1C2=CC(=O)C3=C(C=C(C=C3O2)O)O)O)O
Structural Properties:
Molecular Formula: C15H10O6
Molecular Weight: 286.236
Pharmacophore Features:
Number of bond donors: 4
Number of bond acceptors: 6
Number of atoms different from hydrogen: 21
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Oberdö,rster E, Clay MA, Cottam DM, Wilmot FA, McLachlan JA, Milner MJ. 2001. Common phytochemicals are ecdysteroid agonists and antagonists: a possible evolutionary link between vertebrate and invertebrate steroid hormones. J Steroid Biochem Mol Biol 77(4-5):229-238, DOI: 10.1016/S0960-0760(01)00067-X.
Scippo ML, Argiris C, Van De Weerdt C, Muller M, Willemsen P, Martial J, Maghuin-Rogister G. 2004. Recombinant human estrogen, androgen and progesterone receptors for detection of potential endocrine disruptors. Analytical & Bioanalytical Chemistry 378(3).
External Links

2D-structure
3D-structure