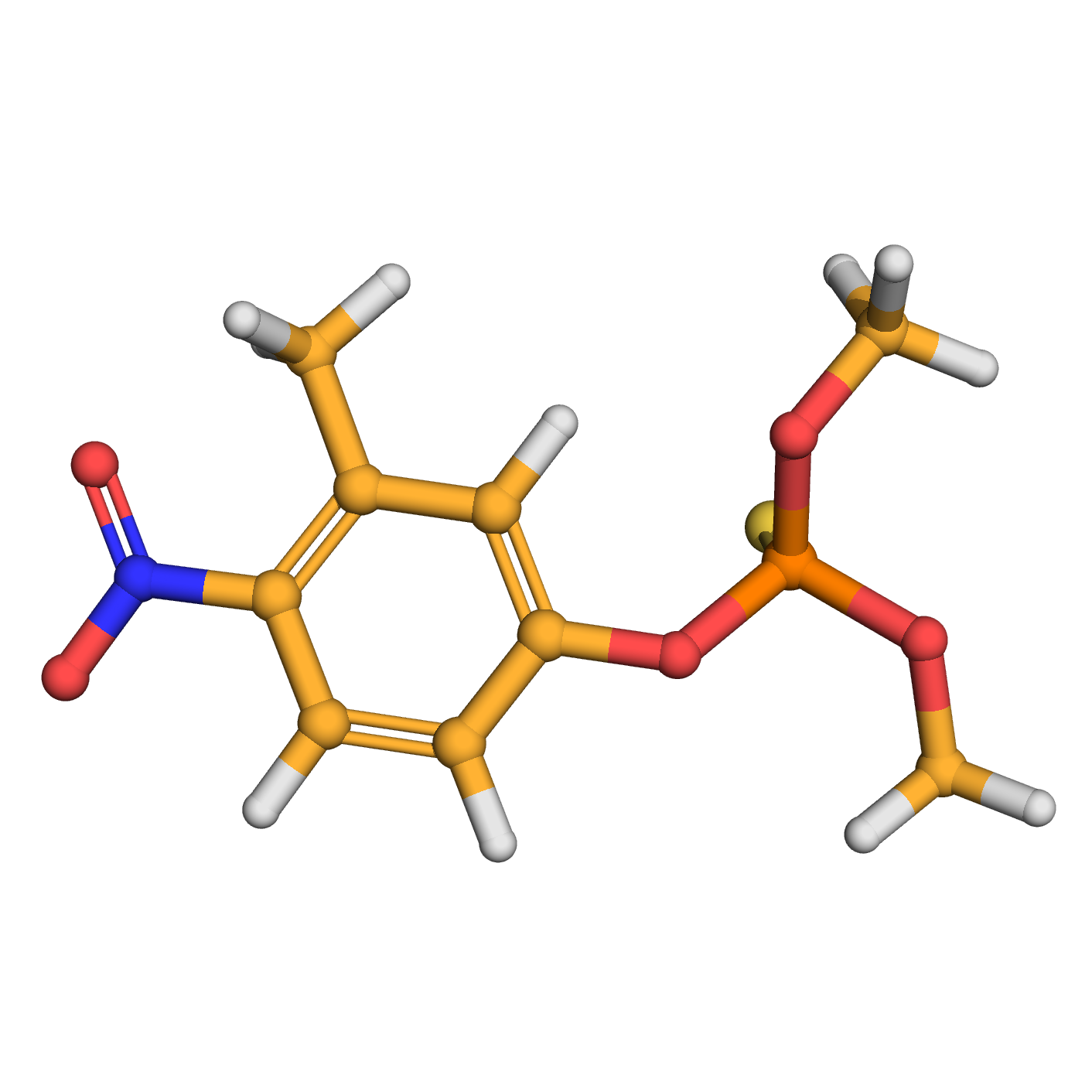
fenitrothion
Synonyms: "phenitrothion", "sumithion", "methylnitrophos", "oleosumifene", "folithion", "metathion", "nitrophos", "ovadofos", "owadofos"
Source: fenitrothion is found in a variety of commercial insecticides, as active ingredient.
Identifiers:
IUPAC Name: dimethoxy-(3-methyl-4-nitrophenoxy)-sulfanylidene-&lambda,5-phosphane
CAS Number: 122-14-5
PubChem ID: 31200
InChiKey: ZNOLGFHPUIJIMJ-UHFFFAOYSA-N
Canonical SMILES: CC1=C(C=CC(=C1)OP(=S)(OC)OC)[N+](=O)[O-]
Structural Properties:
Molecular Formula: C9H12NO5PS
Molecular Weight: 277.234
Pharmacophore Features:
Number of bond donors: 0
Number of bond acceptors: 2
Number of atoms different from hydrogen: 17
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Kojima H, Katsura E, Takeuchi S, Niiyama K, Kobayashi K. 2004. Screening for estrogen and androgen receptor activities in 200 pesticides by in vitro reporter gene assays using Chinese hamster ovary cells. Environ Health Perspect 112(5):524-531.
Tamura H, Maness SC, Reischmann K, Dorman DC, Gray LE, Gaido KW. 2001. Androgen receptor antagonism by the organophosphate insecticide fenitrothion. Toxicol Sci 60(1):56-62.
External Links

2D-structure
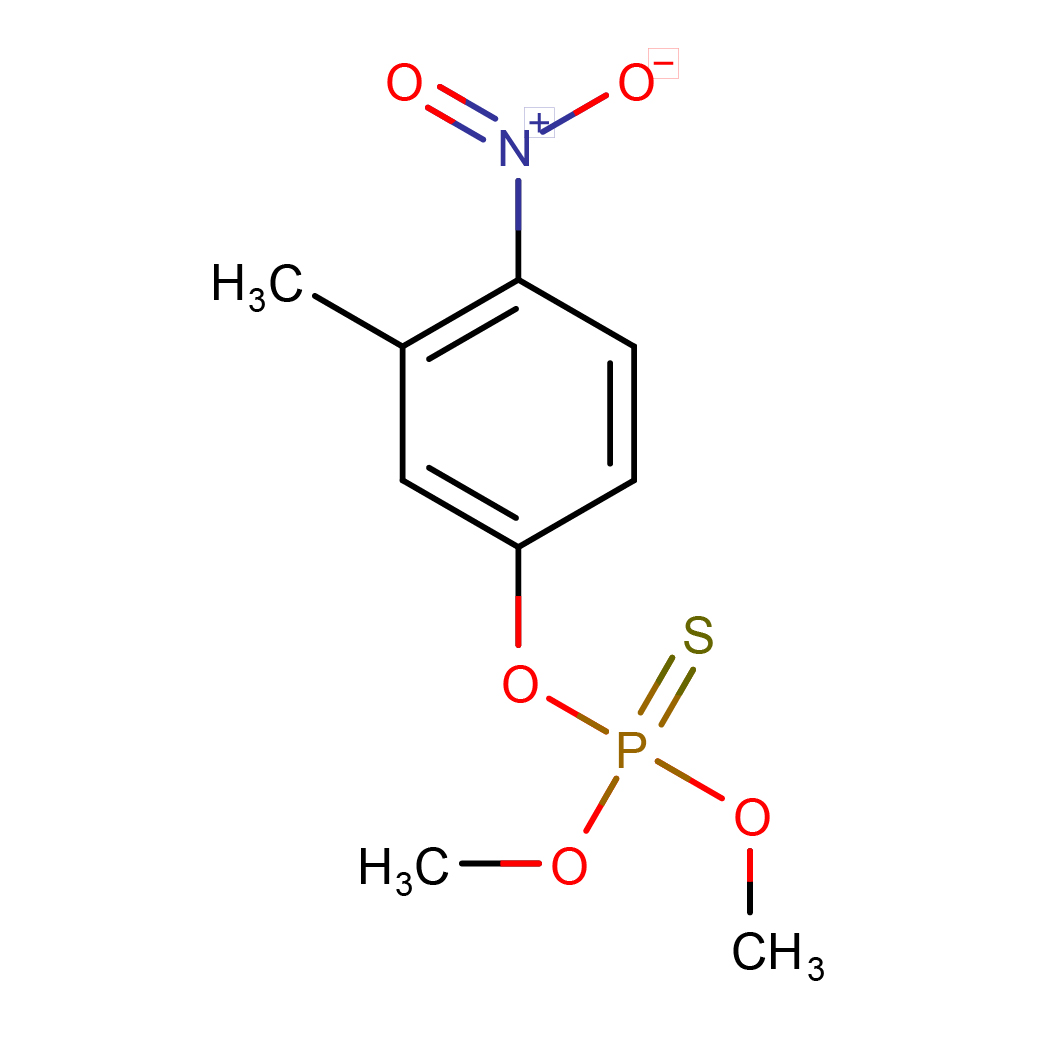
3D-structure