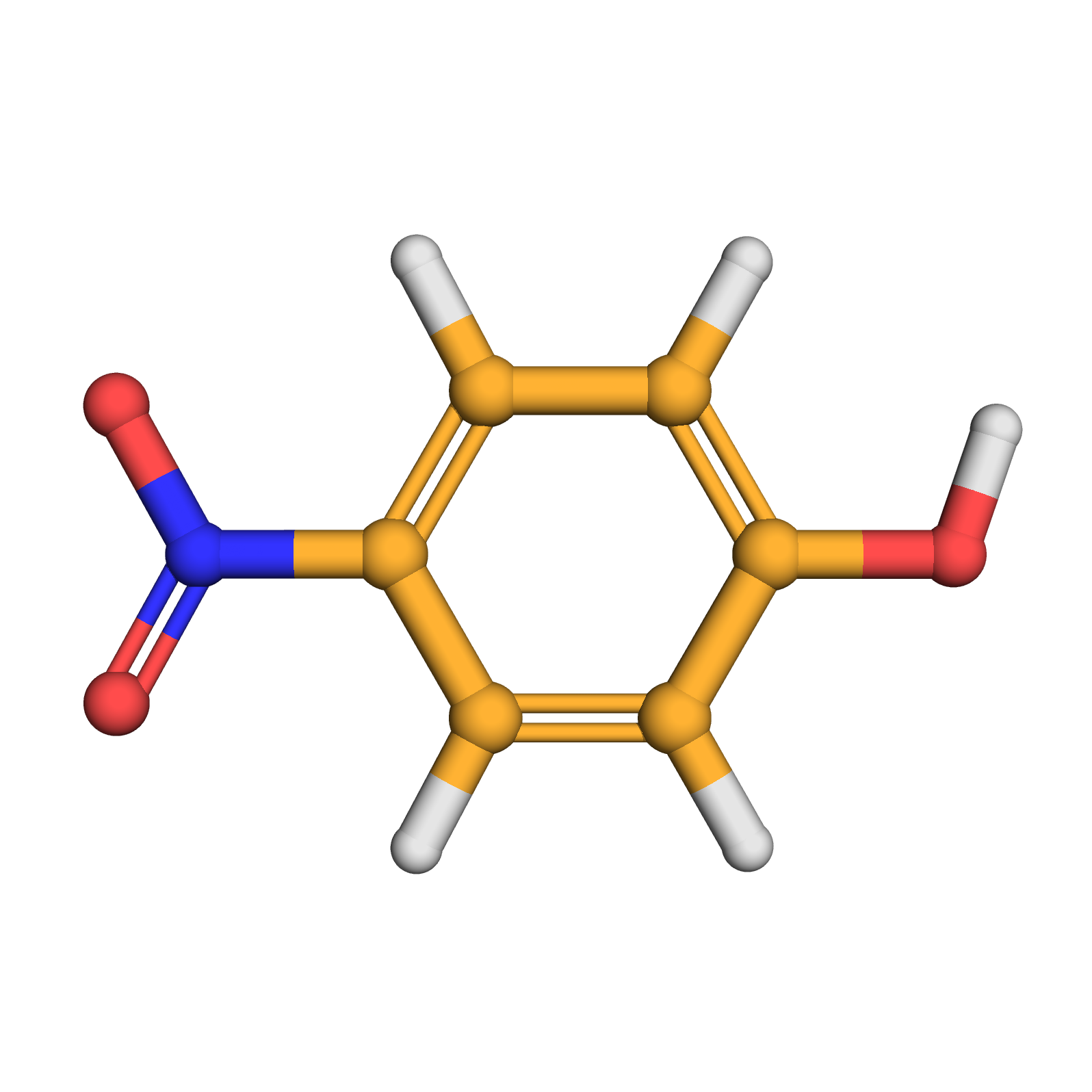
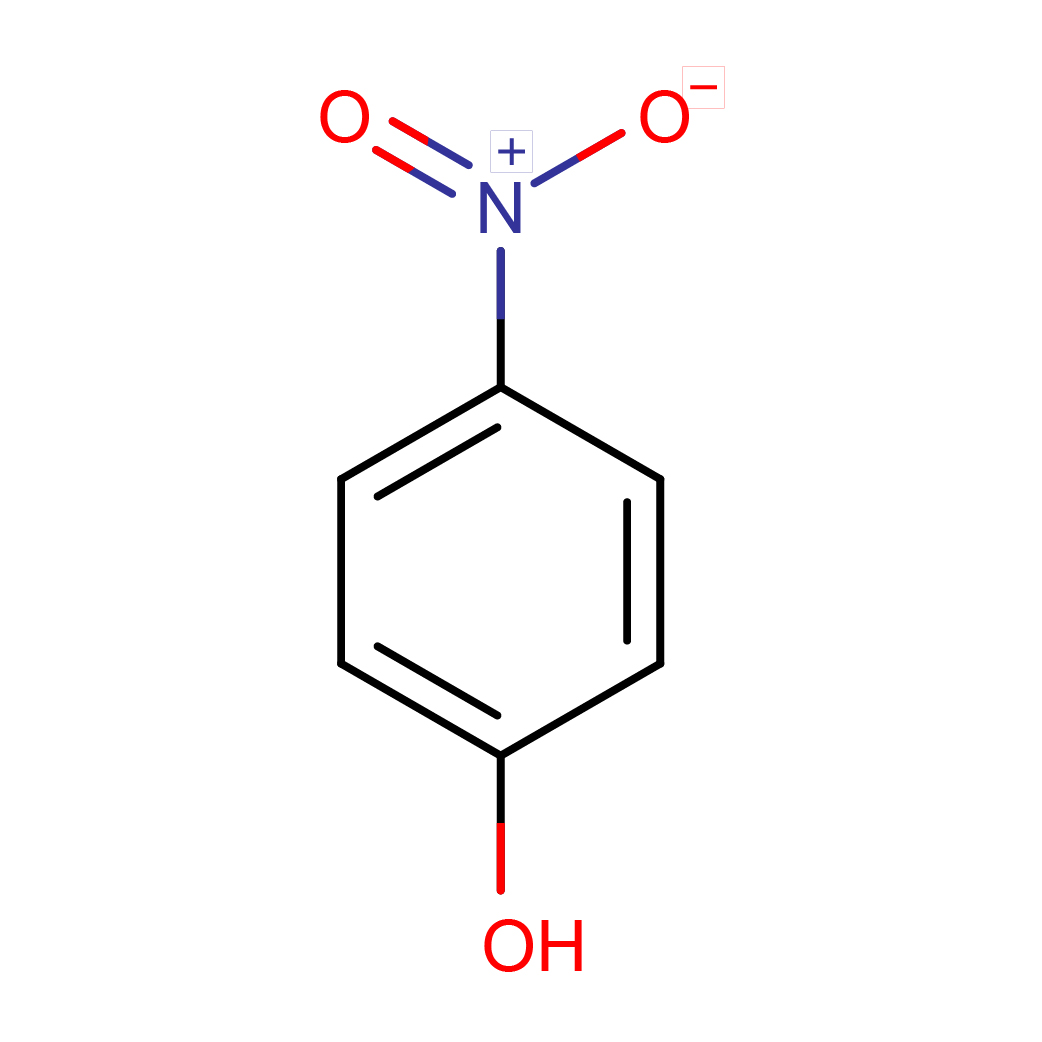
4-nitrophenol
Synonyms: "p-nitrophenol", "niphen", "4-hydroxynitrobenzene", "paranitrophenol", "p-hydroxynitrobenzene"
Source: 4-nitrophenol is used to manufacture drugs, fungicides, insecticides, and dyes and to darken leather.
Identifiers:
IUPAC Name: 4-nitrophenol
CAS Number: 100-02-7
PubChem ID: 980
InChiKey: BTJIUGUIPKRLHP-UHFFFAOYSA-N
Canonical SMILES: C1=CC(=CC=C1[N+](=O)[O-])O
Structural Properties:
Molecular Formula: C6H5NO3
Molecular Weight: 139.109
Pharmacophore Features:
Number of bond donors: 1
Number of bond acceptors: 3
Number of atoms different from hydrogen: 10
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Li C, Taneda S, Suzuki AK, Furuta C, Watanabe G, Taya K. 2006. Estrogenic and anti-androgenic activities of 4-nitrophenol in diesel exhaust particles. Toxicol Appl Pharmacol 217(1):1-6.
Li X, Li C, Suzuki AK, Taneda S, Watanabe G, Taya K. 2009. 4-Nitrophenol isolated from diesel exhaust particles disrupts regulation of reproductive hormones in immature male rats. Endocrine 36(1):98-102.
Mi Y, Zhang C, Li CM, Taneda S, Watanabe G, Suzuki AK, Taya K. 2010. Protective effect of quercetin on the reproductive toxicity of 4-nitrophenol in diesel exhaust particles on male embryonic chickens. J Reprod Dev 56(2):195-199.
External Links



2D-structure
3D-structure