parathion-methyl
Synonyms: "methyl parathion", "methylparathion", "metaphos", "vofatox", "wofatox", "dalf", "parathion methyl", "dimethyl parathion", "metacid 50"
Source: parathion-methyl is a chemical insecticide.
Identifiers:
IUPAC Name: dimethoxy-(4-nitrophenoxy)-sulfanylidene-?5-phosphane
CAS Number: 298-00-0
PubChem ID: 4130
InChiKey: RLBIQVVOMOPOHC-UHFFFAOYSA-N
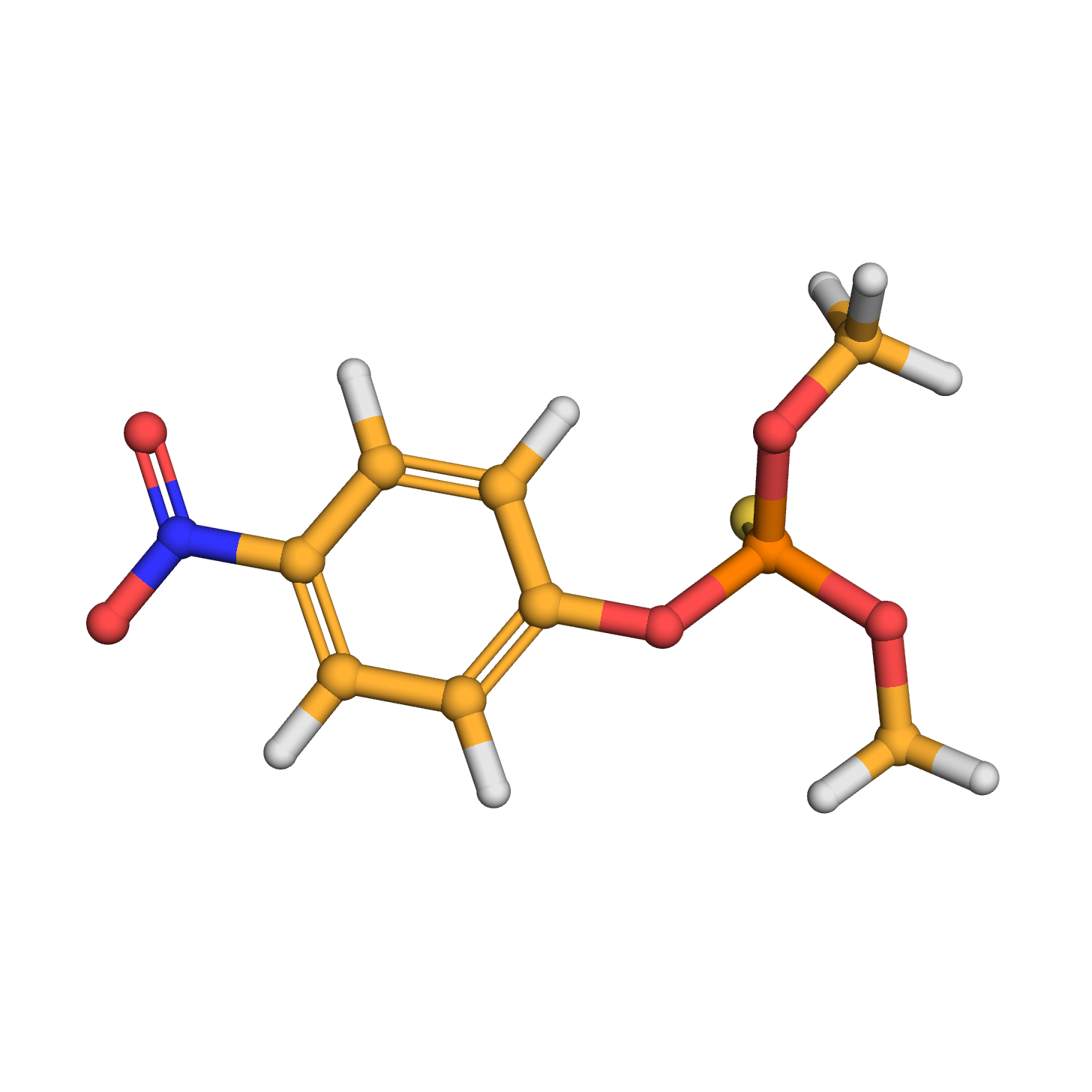
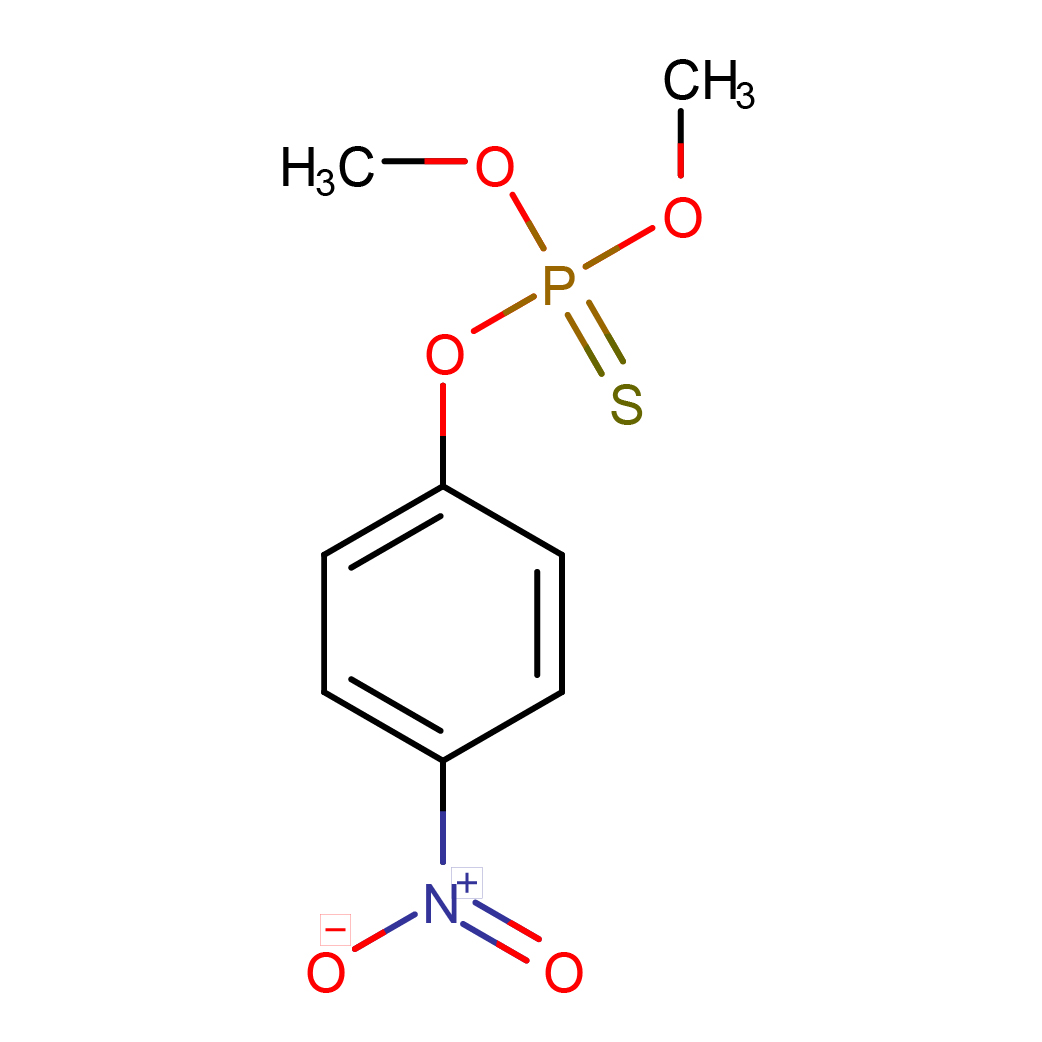
Canonical SMILES: COP(=S)(OC)OC1=CC=C(C=C1)[N+](=O)[O-]
Structural Properties:
Molecular Formula: C8H10NO5PS
Molecular Weight: 263.207
Pharmacophore Features:
Number of bond donors: 0
Number of bond acceptors: 2
Number of atoms different from hydrogen: 16
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Crittenden PL, Carr R, Pruett SB. 1998. Immunotoxicological assessment of methyl parathion in female B6C3F1 mice. J Toxicol Environ Health A 54(1):1-20.Kojima H, Katsura E, Takeuchi S, Niiyama K, Kobayashi K. 2004. Screening for estrogen and androgen receptor activities in 200 pesticides by in vitro reporter gene assays using Chinese hamster ovary cells. Environ Health Perspect 112(5):524-531.
Lukaszewicz-Hussain A, Moniuszko-Jakoniuk J, Pawlowska D. 1985. Blood glucose and insulin concentration in rats subjected to physical exercise in acute poisoning with parathion-methyl. Pol J Pharmacol Pharm 37(5):647-651.
Petit F, Le Goff P, Cravedi JP, Valotaire Y, Pakdel F. 1997. Two complementary bioassays for screening the estrogenic potency of xenobiotics: recombinant yeast for trout estrogen receptor and trout hepatocyte cultures. J Mol Endocrinol 19(3):321-335.
External Links

2D-structure
3D-structure