pendimethalin
Synonyms: "penoxaline", "prowl", "pendimethaline", "accotab", "herbadox", "phenoxalin", "stomp", "tendimethalin", "N-(1-ethylpropyl)-2,6-dinitro-3,4-xylidine", "3,4-Dimethyl-2,6-dinitro-N-(1-ethylpropyl)aniline"
Source: pendimethalin is a selective herbicide used to control most annual grasses and certain broadleaf weeds in field corn, potatoes, rice, cotton, soybeans, tobacco, peanuts and sunflowers.
Identifiers:
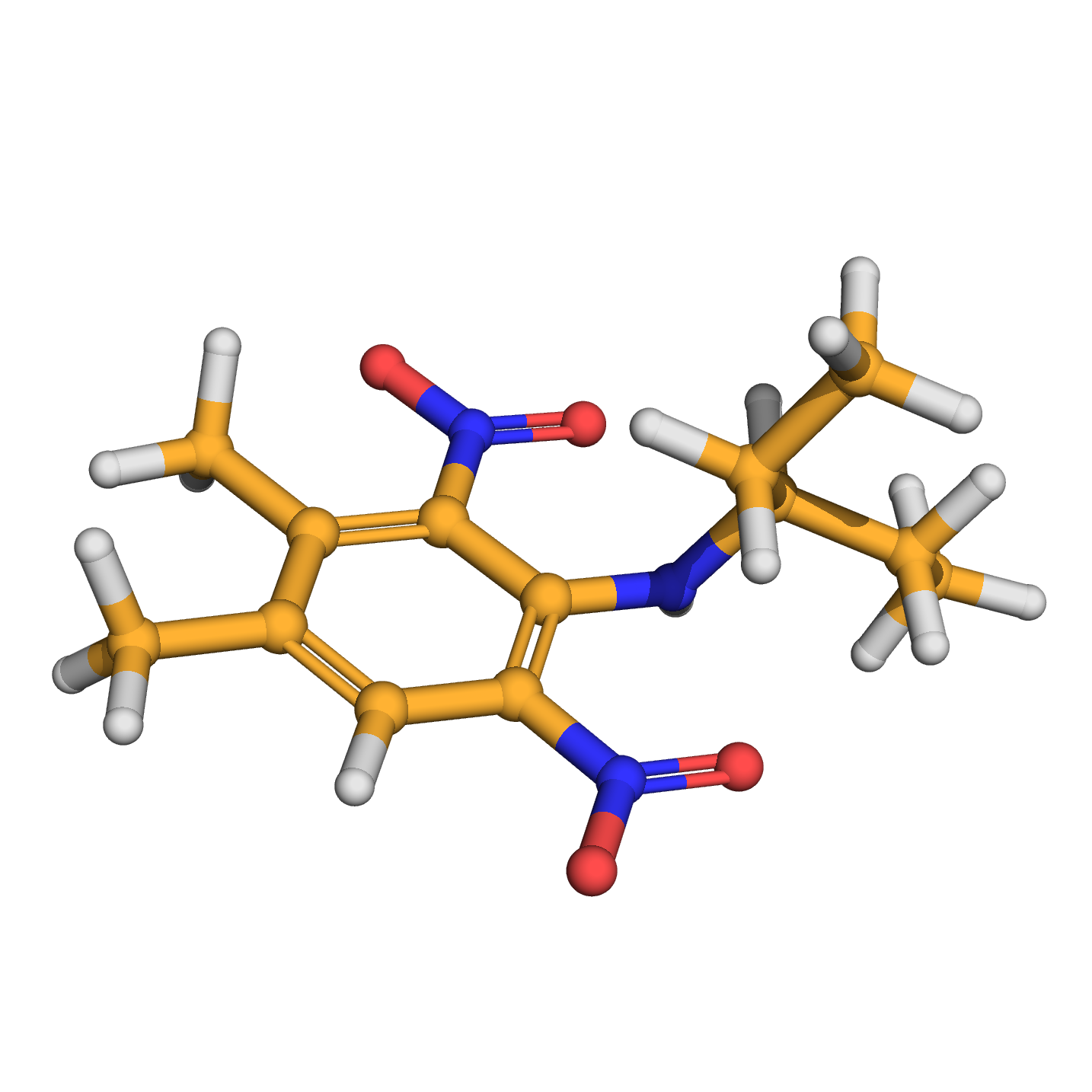
IUPAC Name: 3,4-dimethyl-2,6-dinitro-N-pentan-3-ylaniline
CAS Number: 40487-42-1
PubChem ID: 38479
InChiKey: CHIFOSRWCNZCFN-UHFFFAOYSA-N
Canonical SMILES: CCC(CC)NC1=C(C=C(C(=C1[N+](=O)[O-])C)C)[N+](=O)[O-]
Structural Properties:
Molecular Formula: C13H19N3O4
Molecular Weight: 281.308
Pharmacophore Features:
Number of bond donors: 1
Number of bond acceptors: 5
Number of atoms different from hydrogen: 20
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Hurley PM, Hill RN, Whiting RJ. 1998. Mode of carcinogenic action of pesticides inducing thyroid follicular cell tumors in rodents [review]. Environ Health Perspect 106(8):437-445.
Kojima H, Katsura E, Takeuchi S, Niiyama K, Kobayashi K. 2004. Screening for estrogen and androgen receptor activities in 200 pesticides by in vitro reporter gene assays using Chinese hamster ovary cells. Environ Health Perspect 112(5):524-531.
External Links


2D-structure
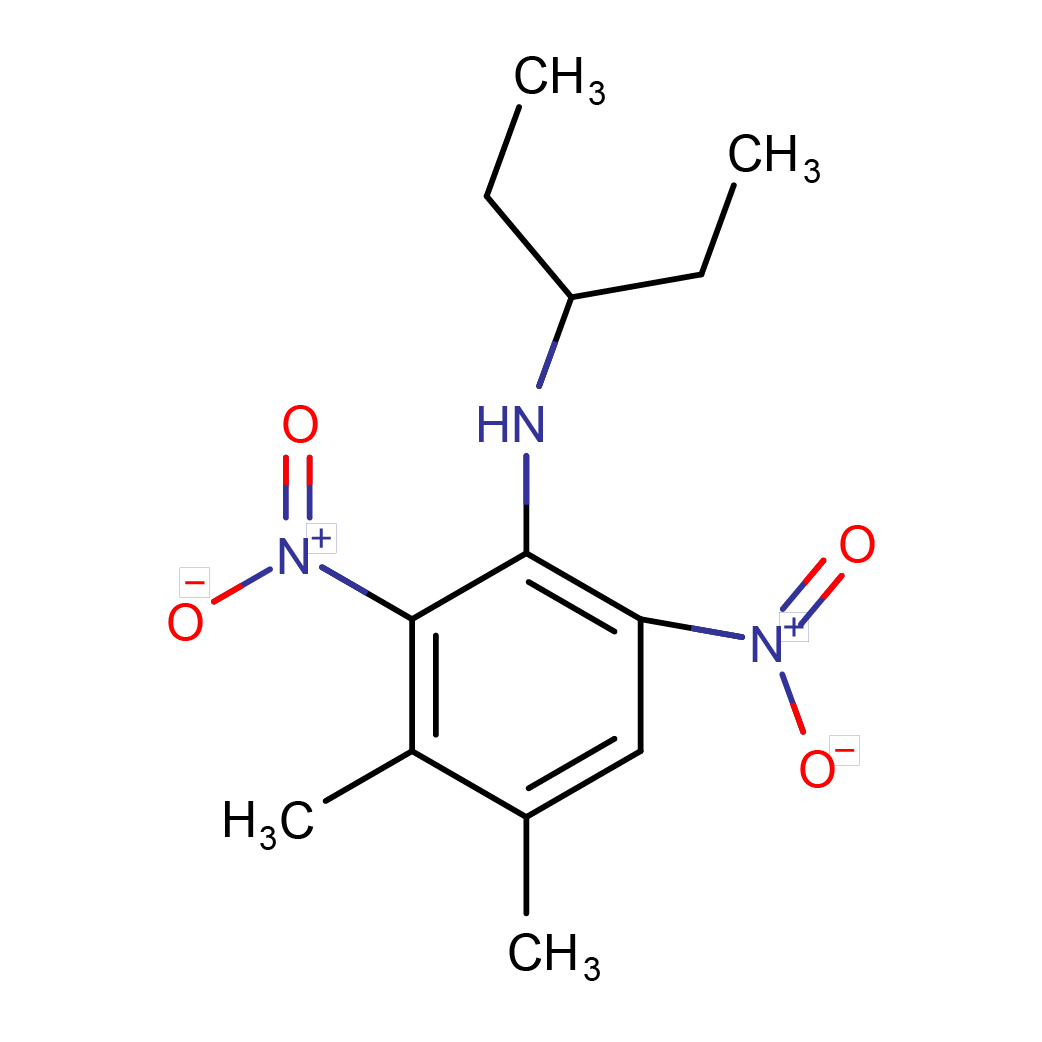
3D-structure