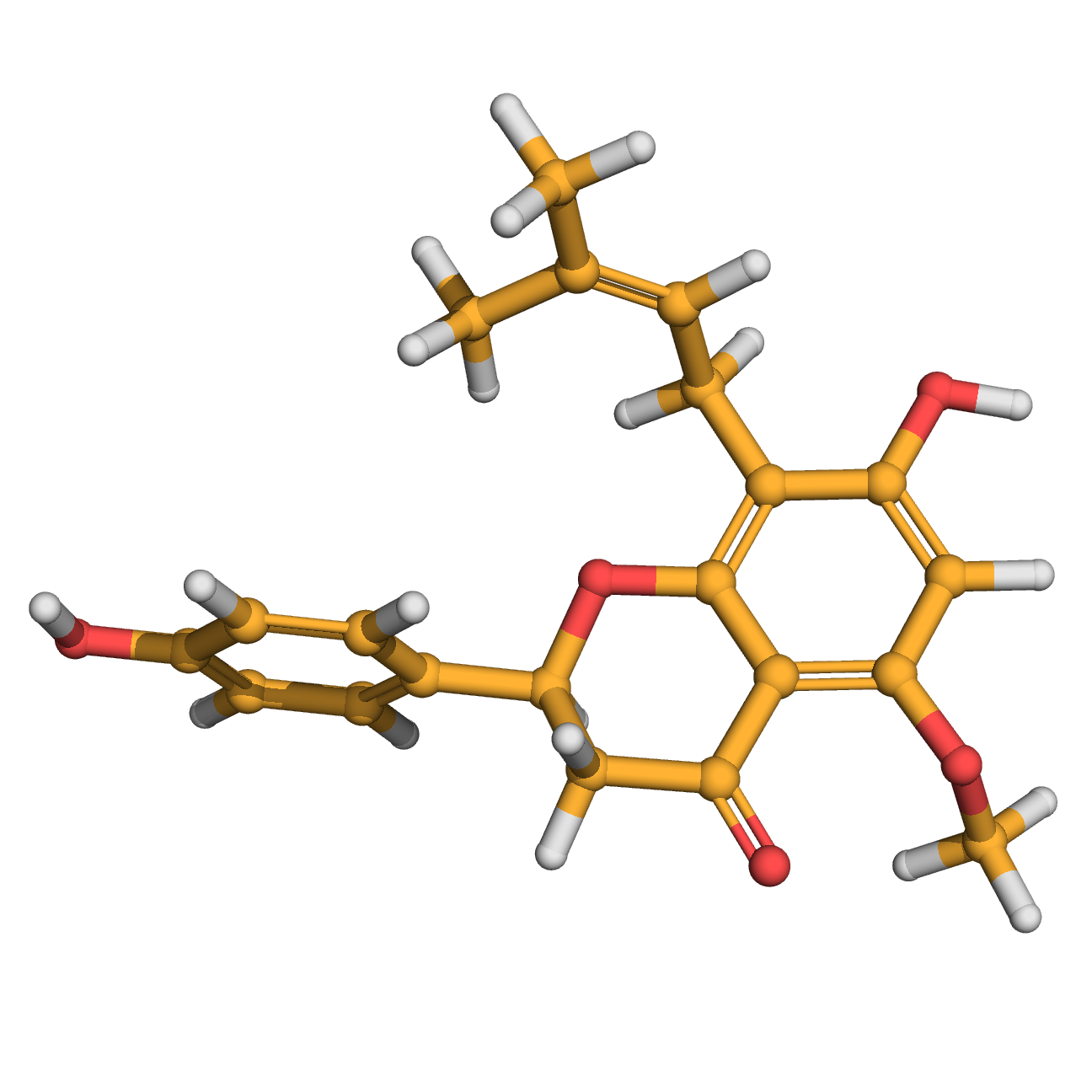
isoxanthohumol
Synonyms: "7-hydroxy-2-(4-hydroxyphenyl)-5-methoxy-8-(3-methylbut-2-enyl)-2,3-dihydrochromen-4-one", "7-hydroxy-2-(4-hydroxyphenyl)-5-methoxy-8-(3-methyl-2-butenyl)-2,3-dihydro-4H-chromen-4-one"
Source: isoxanthohumol is the most abundant and an important prenylated flavonoid present in hopped beers.
Identifiers:
IUPAC Name: 7-hydroxy-2-(4-hydroxyphenyl)-5-methoxy-8-(3-methylbut-2-enyl)-2,3-dihydrochromen-4-one
CAS Number: 70872-29-6
PubChem ID: 513197
InChiKey: YKGCBLWILMDSAV-UHFFFAOYSA-N
Canonical SMILES: CC(=CCC1=C(C=C(C2=C1OC(CC2=O)C3=CC=C(C=C3)O)OC)O)C
Structural Properties:
Molecular Formula: C21H22O5
Molecular Weight: 354.396
Pharmacophore Features:
Number of bond donors: 2
Number of bond acceptors: 5
Number of atoms different from hydrogen: 26
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Coldham NG, Sauer MJ. 2001. Identification, quantitation and biological activity of phytoestrogens in a dietary supplement for breast enhancement. Food & Chemical Toxicology 39(12):1211-1224.
Izzo G, Soder O, Svechnikov K. 2011. The prenylflavonoid phytoestrogens 8-prenylnaringenin and isoxanthohumol diferentially suppress steroidogenesis in rat Leydig cells in ontogenesis. J Appl Toxicol 31(6):589-594.
External Links


2D-structure
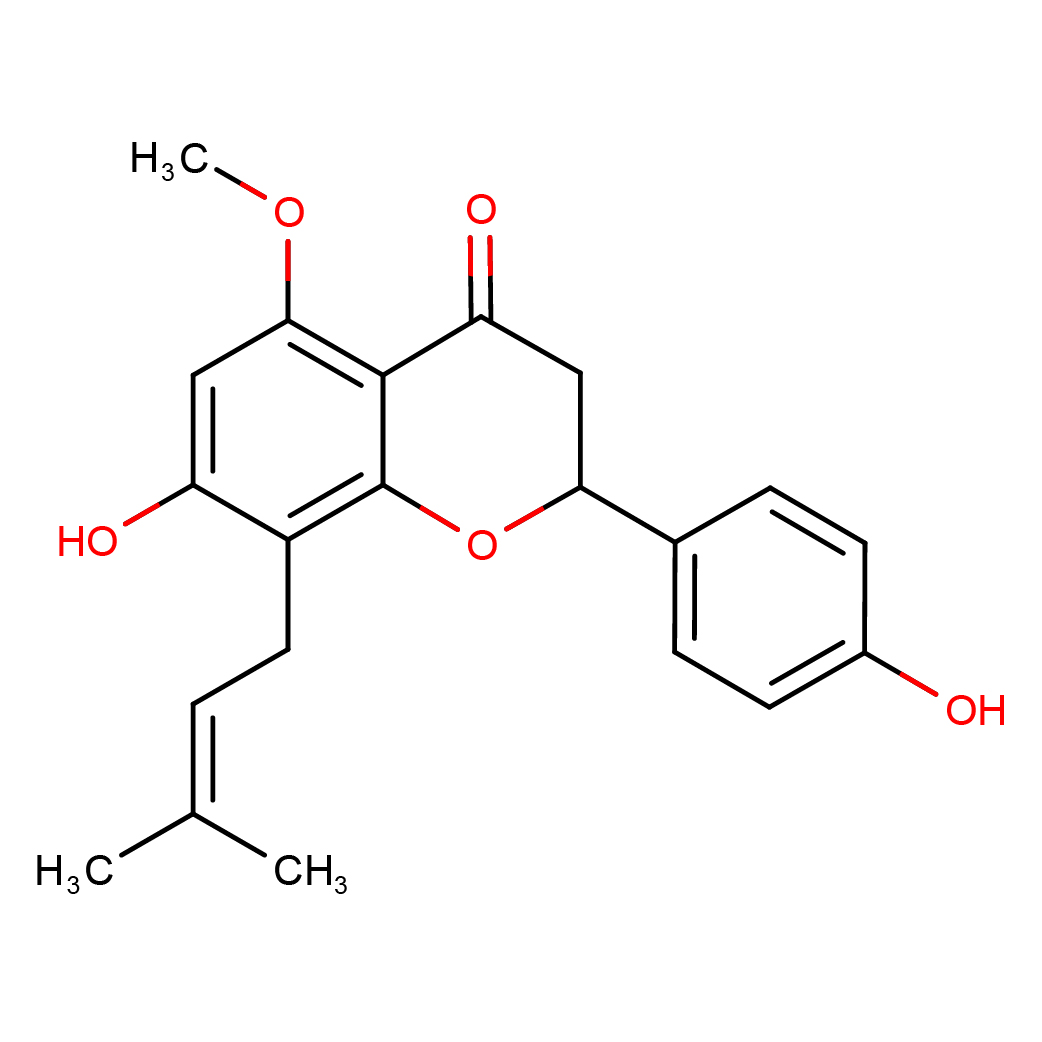
3D-structure