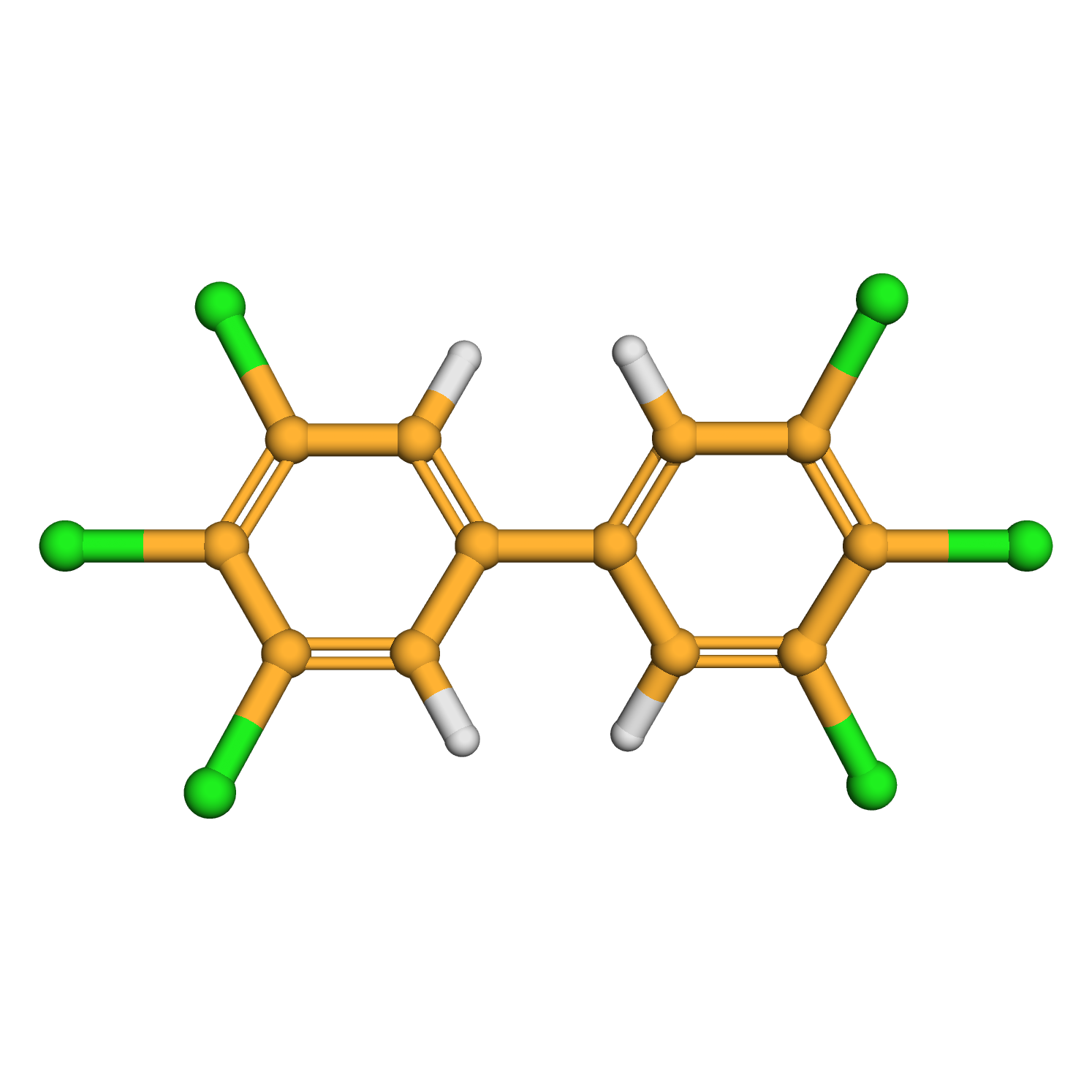
3,3',4,4',5,5'-hexachlorobiphenyl
Synonyms: "PCB 169", "3,3',4,4',5,5'-hexachlorobiphenyl"
Source: 3,3',4,4',5,5'-hexachlorobiphenyl is a member of the polychlorinated biphenyl (PCB), a group of industrial chemicals used as dielectrics, coolants and lubricants in electrical equipment. PCBs contaminate water by leaching from landfills or other waste deposits.
Identifiers:
IUPAC Name: 1,2,3-trichloro-5-(3,4,5-trichlorophenyl)benzene
CAS Number: 32774-16-6
PubChem ID: 36231
InChiKey: ZHLICBPIXDOFFG-UHFFFAOYSA-N
Canonical SMILES: C1=C(C=C(C(=C1Cl)Cl)Cl)C2=CC(=C(C(=C2)Cl)Cl)Cl
Structural Properties:
Molecular Formula: C12H4Cl6
Molecular Weight: 360.878
Pharmacophore Features:
Number of bond donors: 0
Number of bond acceptors: 0
Number of atoms different from hydrogen: 18
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Krishnan V, Safe S. 1993. Polychlorinated biphenyls (PCBs), dibenzo-p-dioxins (PCDDs), and dibenzofurans (PCDFs) as antiestrogens in MCF-7 human breast cancer cells: quantitative structure-activity relationships. Toxicol Appl Pharmacol 120(1):55-61.
Patnode KA, Curtis LR. 1994. 2,2',4,4',5,5'- and 3,3',4,4',5,5'-hexachlorobiphenyl alteration of uterine progesterone and estrogen receptors coincides with embryotoxicity in mink (Mustela vison). Toxicol Appl Pharmacol 127(1):9-18.
External Links



2D-structure
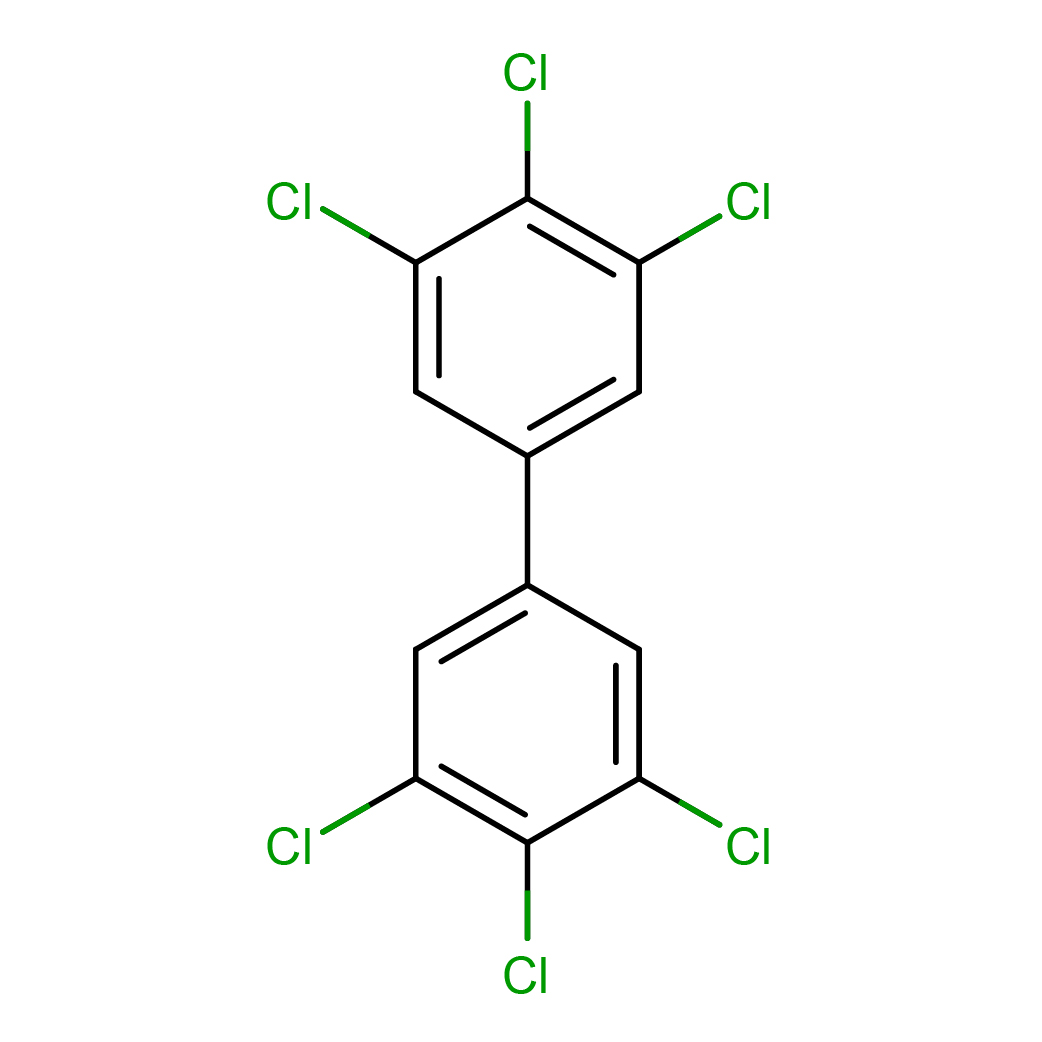
3D-structure