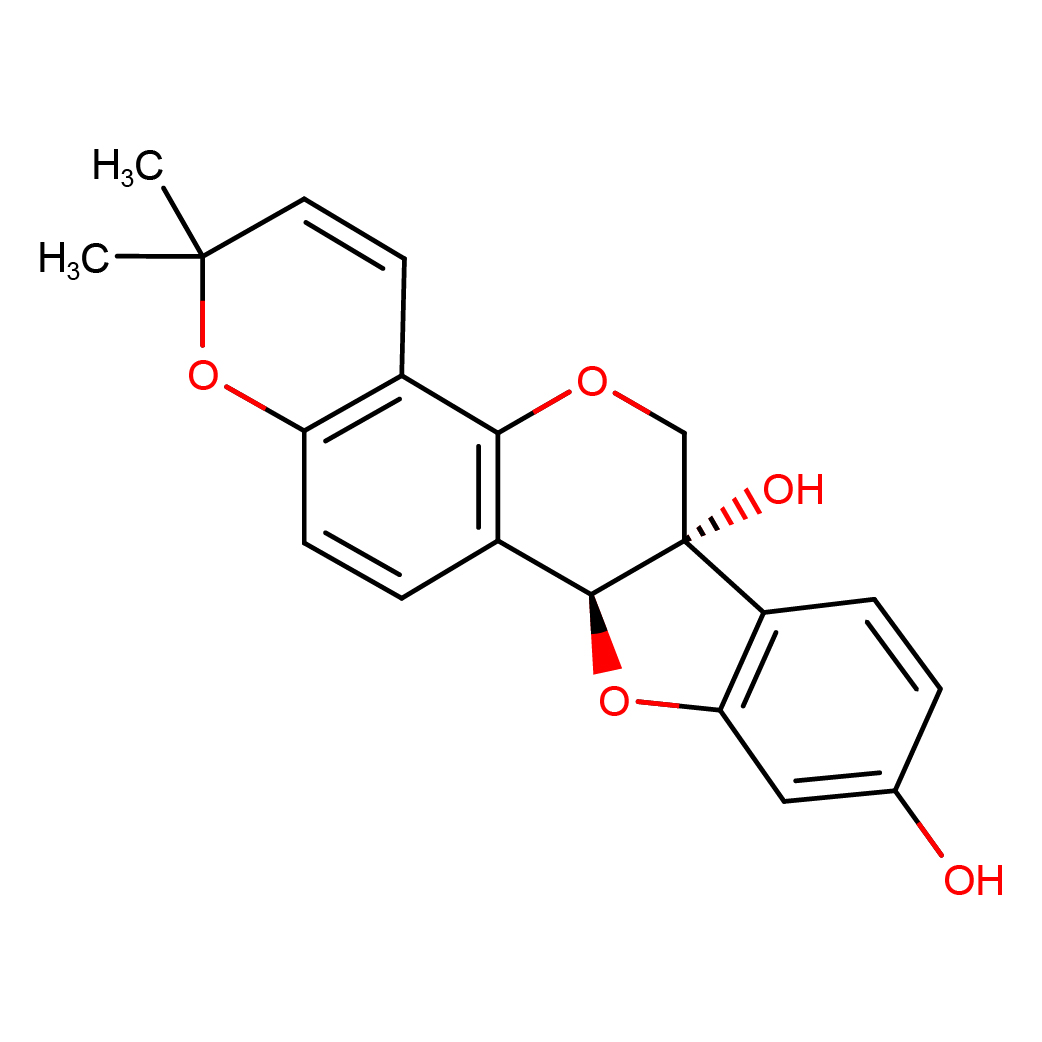
glyceollin I
Synonyms: "(-)-glyceollin I", "(6aS,11aS)-2,2-dimethyl-2H,6H-[1]benzofuro[3,2-c]pyrano[2,3-h]chromene-6a,9(11aH)-diol"
Source: glyceollin I is a glyceollin, a type of prenylated pterocarpan. It is a phytoalexin found in the soybean.
Identifiers:
IUPAC Name: (6aS,11aS)-2,2-dimethyl-2H,6H-[1]benzofuro[3,2?c]pyrano[2,3?h]chromene-6a,9(11aH)-diol
CAS Number: 57103-57-8
PubChem ID: 162807
InChiKey: YIFYYPKWOQSCRI-AZUAARDMSA-N
Canonical SMILES: CC1(C=CC2=C(O1)C=CC3=C2OCC4(C3OC5=C4C=CC(=C5)O)O)C
Structural Properties:
Molecular Formula: C20H18O5
Molecular Weight: 338.354
Pharmacophore Features:
Number of bond donors: 2
Number of bond acceptors: 5
Number of atoms different from hydrogen: 25
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Burow ME, Boue SM, Collins-Burow BM, Melnik LI, Duong BN, Carter-Wientjes CH, Li S, Wiese TE, Cleveland TE, McLachlan JA. 2001. Phytochemical glyceollins, isolated from soy, mediate antihormonal effects through estrogen receptor alpha and beta. J Clin Endocrinol Metab 86(4):1750-1758.
External Links

2D-structure
3D-structure