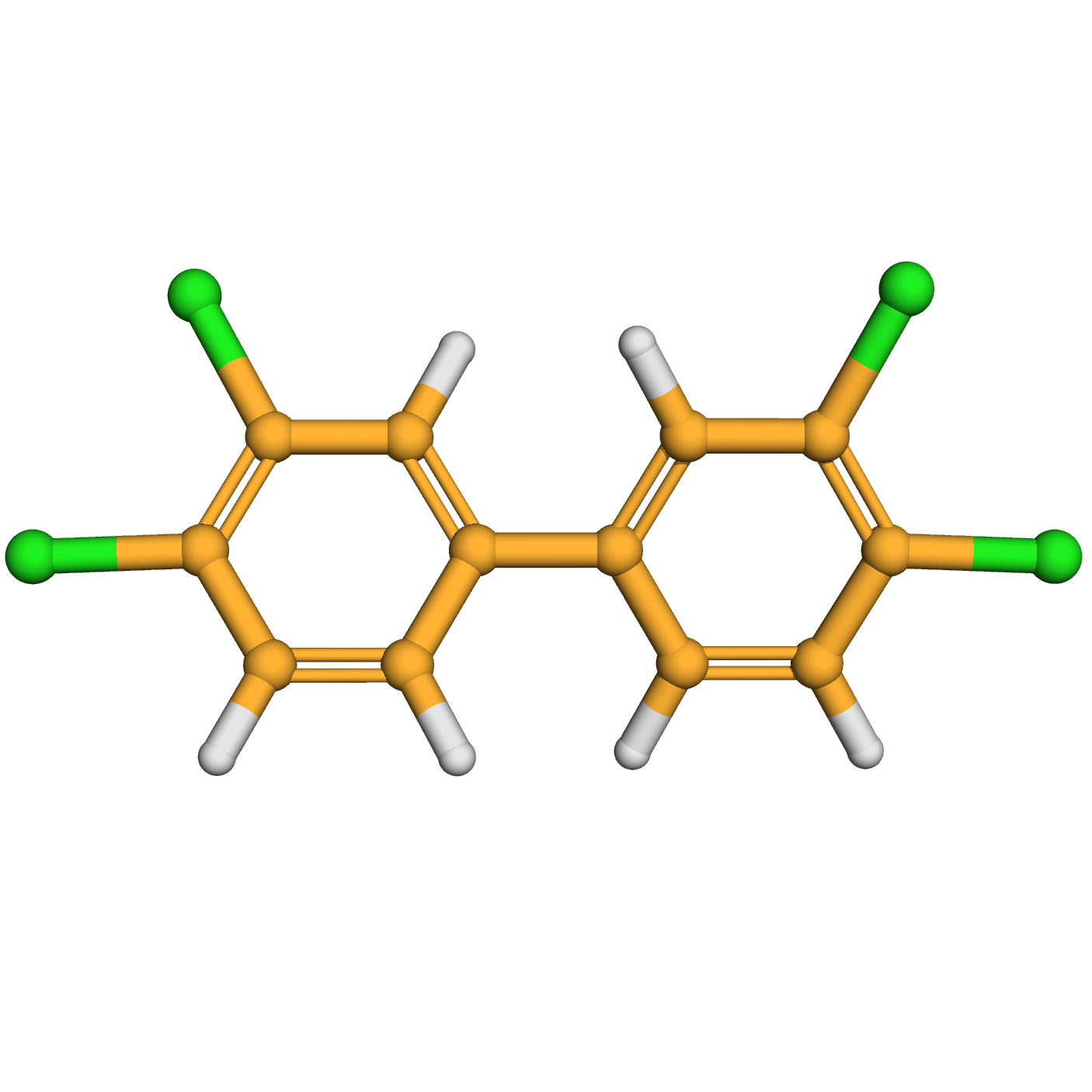
3,3',4,4'-tetrachlorobiphenyl
Synonyms: "3,4,3',4'-tetrachlorobiphenyl", "PCB 77", "4-CB"
Source: 3,3',4,4'-tetrachlorobiphenyl is a member of the polychlorinated biphenyl (PCB), a group of industrial chemicals used as dielectrics, coolants and lubricants in electrical equipment. PCBs contaminate water by leaching from landfills or other waste deposits.
Identifiers:
IUPAC Name: 1,2-dichloro-4-(3,4-dichlorophenyl)benzene
CAS Number: 32598-13-3
PubChem ID: 36187
InChiKey: UQMGJOKDKOLIDP-UHFFFAOYSA-N
Canonical SMILES: C1=CC(=C(C=C1C2=CC(=C(C=C2)Cl)Cl)Cl)Cl
Structural Properties:
Molecular Formula: C12H6Cl4
Molecular Weight: 291.988
Pharmacophore Features:
Number of bond donors: 0
Number of bond acceptors: 0
Number of atoms different from hydrogen: 16
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Durham SK, Brouwer A. 1990. 3,4,3',4'-Tetrachlorobiphenyl distribution and induced effects in the rat adrenal gland: Localization in the zona fasciculata. Lab Invest 62(2):232-239.
Schantz SL, Seo B-W, Moshtaghian J, Amin S. 1997. Developmental exposure to polychlorinated biphenyls or dioxin: do changes in thyroid function mediate effects on spatial learning? Am Zool 37(4):399-408.
Van den Berg KJ, van Raaij JAGM, Bragt PC, Notten WRF. 1991. Interactions of halogenated industrial chemicals with transthyretin and effects on thyroid hormone levels in vivo. Arch Toxicol 65(1):15-19.
van den Berg KJ, Zurcher C, Brouwer A. 1988. Effects of 3,4,3',4'-tetrachlorobiphenyl on thyroid function and histology in marmoset monkeys. Toxicol Lett 41(1):77-86.
External Links



2D-structure
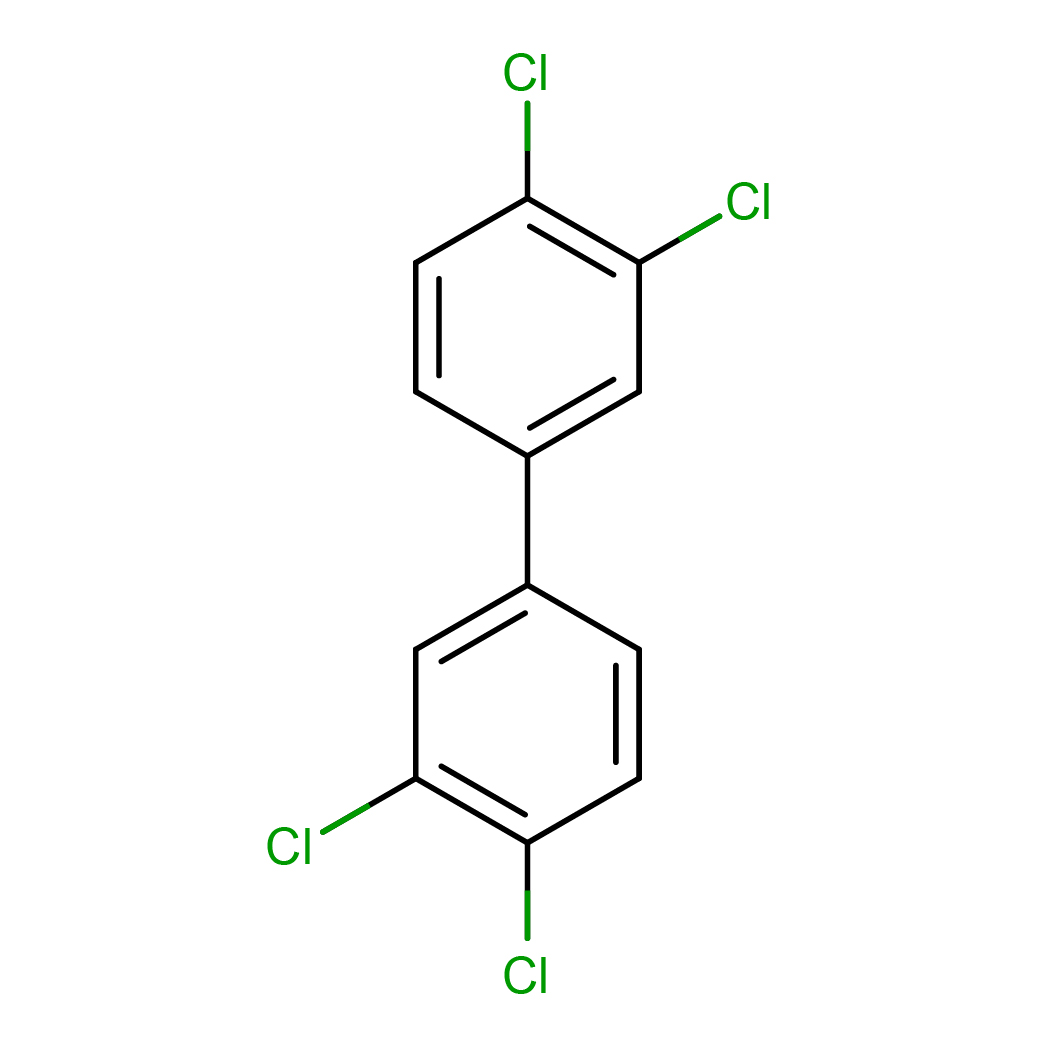
3D-structure