epichlorohydrin
Synonyms: "epichlorhydrin", "2-(chloromethyl)oxirane", "1-chloro-2,3-epoxypropane", "glycidyl chloride", "chloropropylene oxide"
Source: epichlorohydrin is mainly used in the production of epoxy resins.
Identifiers:
IUPAC Name: 2-(chloromethyl)oxirane
CAS Number: 106-89-8
PubChem ID: 7835
InChiKey: BRLQWZUYTZBJKN-UHFFFAOYSA-N
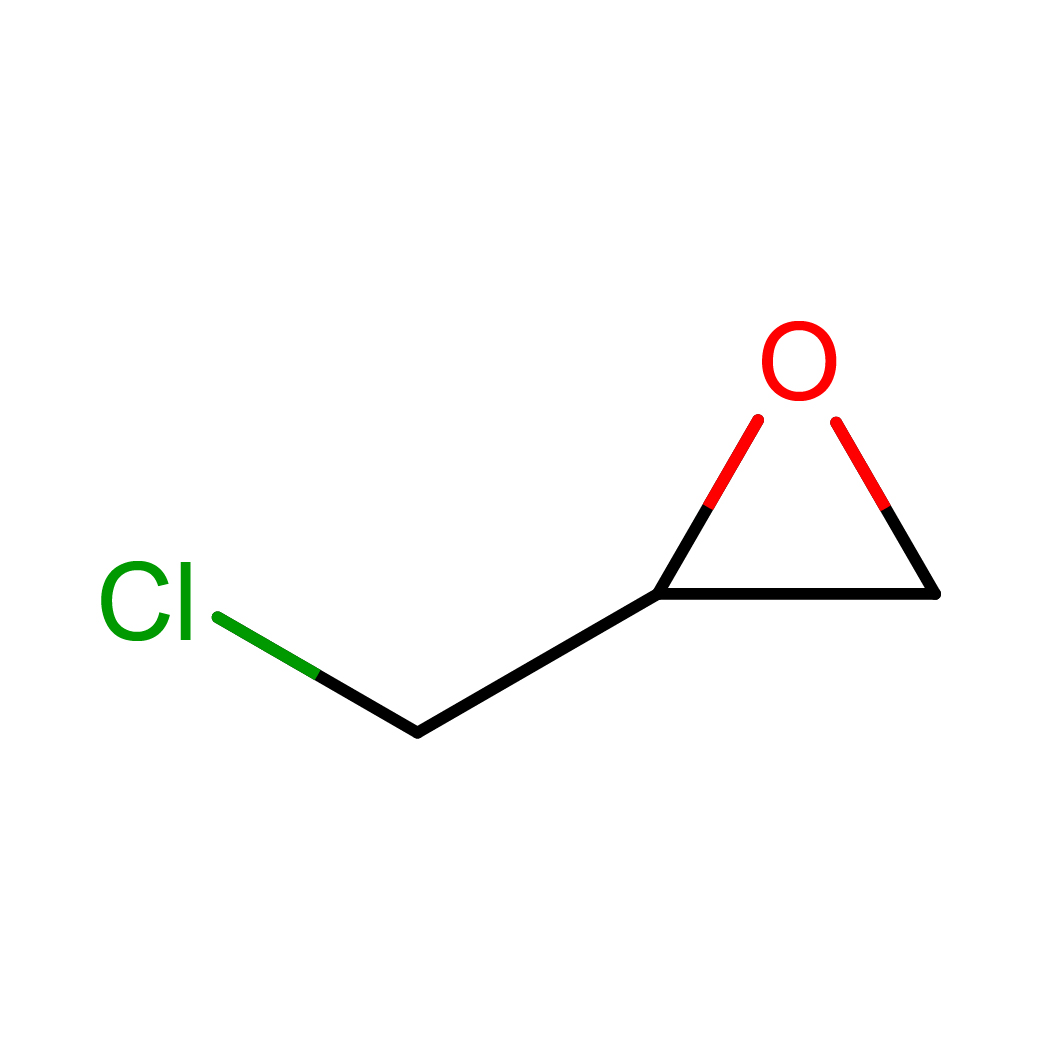
Canonical SMILES: C1C(O1)CCl
Structural Properties:
Molecular Formula: C3H5ClO
Molecular Weight: 92.524
Pharmacophore Features:
Number of bond donors: 0
Number of bond acceptors: 1
Number of atoms different from hydrogen: 5
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Cooper ERA, Jones AR, Jackson H. 1974. Effects of alpha-chlorohydrin and related compounds on the reproductive organs and fertility of the male rat. J Reprod Fertil 38(2):379-386.
John JA, Quast JF, Murray FJ, Calhoun LG, Staples RE. 1983. Inhalation toxicity of epichlorohydrin: effects on fertility in rats and rabbits. Toxicol Appl Pharmacol 68(3):415-423.
Kluwe WM, Gupta BN, Lamb JC 4th. 1983. The comparative effects of 1,2-dibromo-3-chloropropane (DBCP) and its metabolites, 3-chloro-1,2-propaneoxide (epichlorohydrin), 3-chloro-1,2- propanediol (alphachlorohydrin), and oxalic acid, on the urogenital system of male rats. Toxicol Appl Pharmacol 70(1):67-86.
Slott VL, Suarez JD, Simmons JE, Perreault SD. 1990. Acute inhalation exposure to epichlorohydrin transiently decreases rat sperm velocity. Fundam Appl Toxicol 15(3):597-606.
Toth GP, Zenick H, Smith MK. 1989. Effects of epichlorohydrin on male and female reproduction in Long- Evans rats. Fundam Appl Toxicol 13(1):16-25.
External Links


2D-structure
3D-structure