nicotine
Synonyms: "(-)-nicotine", "(S)-nicotine", "nicoderm", "nicotrol", "nicoderm Cq", "(S)-3-(1-methylpyrrolidin-2-yl)pyridine", "micotine", "(S)-(-)-nicotine"
Source: nicotine is a potent parasympathomimetic alkaloid found in the nightshade family of plants (Solanaceae) and a stimulant drug. Nicotine is used in cigarettes and is the main reason people become addicted. It is also used medicinally in nicotine replacement therapies such as nicotine patch and nicotine gum.
Identifiers:
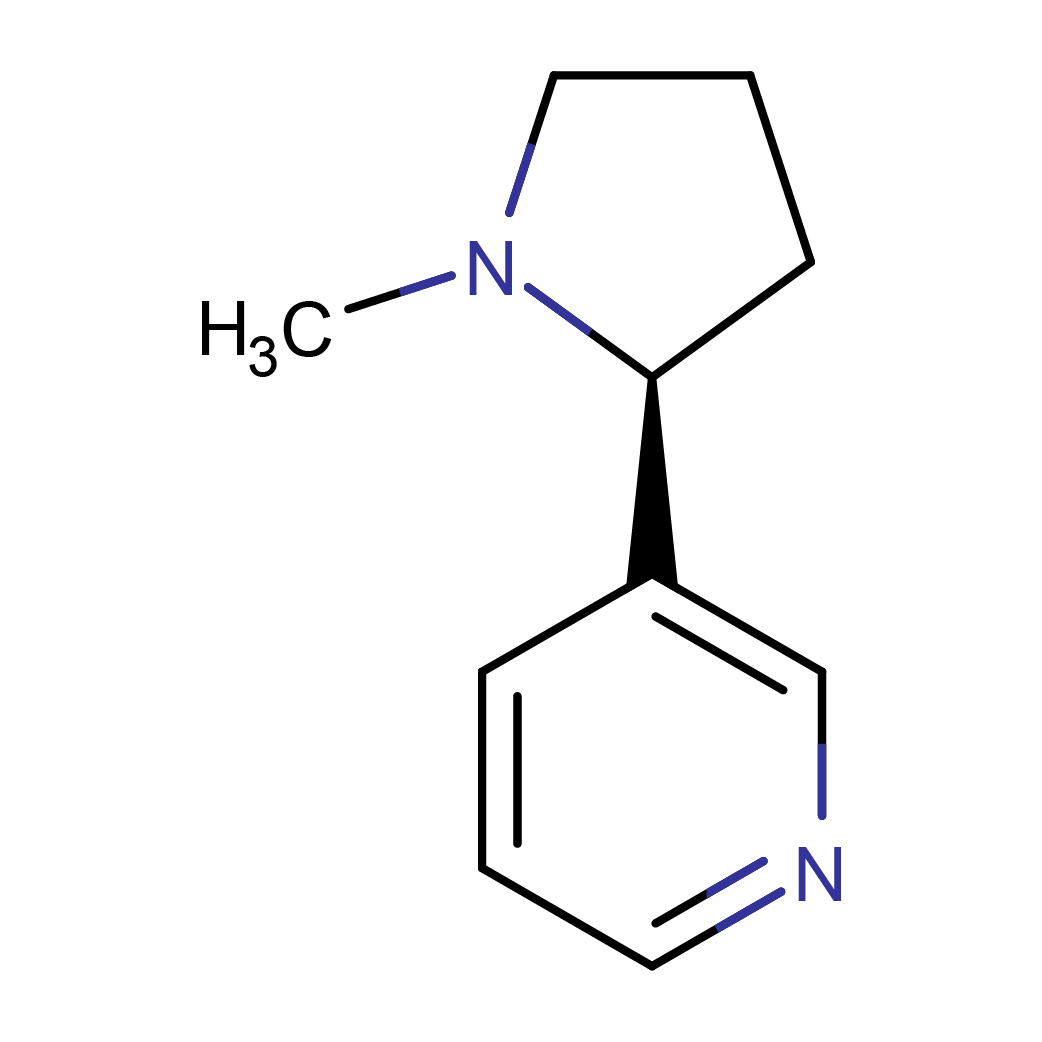
IUPAC Name: 3-[(2S)-1-methylpyrrolidin-2-yl]pyridine
CAS Number: 54-11-5
PubChem ID: 89594
InChiKey: SNICXCGAKADSCV-JTQLQIEISA-N
Canonical SMILES: CN1CCCC1C2=CN=CC=C2
Structural Properties:
Molecular Formula: C10H14N2
Molecular Weight: 162.232
Pharmacophore Features:
Number of bond donors: 0
Number of bond acceptors: 2
Number of atoms different from hydrogen: 12
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Gyekis J, Anthony K, Foreman JE, Klein LC, Vandenbergh DJ. 2010. Perinatal nicotine exposure delays genital development in mice. Reprod Toxicol 29(3):378-30.
Izrael M, Van der Zee EA, Slotkin TA, Yanai J. 2004. Cholinergic synaptic signaling mechanisms underlying behavioral teratogenicity: effects of nicotine, chlorpyrifos, and heroin converge on protein kinase C translocation in the intermedial part of the hyperstriatum ventrale and on imprinting behavior in an avian model. J Neurosci Res 78(4):499-507.
Qiao D, Seidler FJ, Slotkin TA. 2005. Oxidative mechanisms contributing to the developmental neurotoxicity of nicotine and chlorpyrifos. Toxicol Appl Pharmacol 206(1):17-26.
External Links



2D-structure
3D-structure