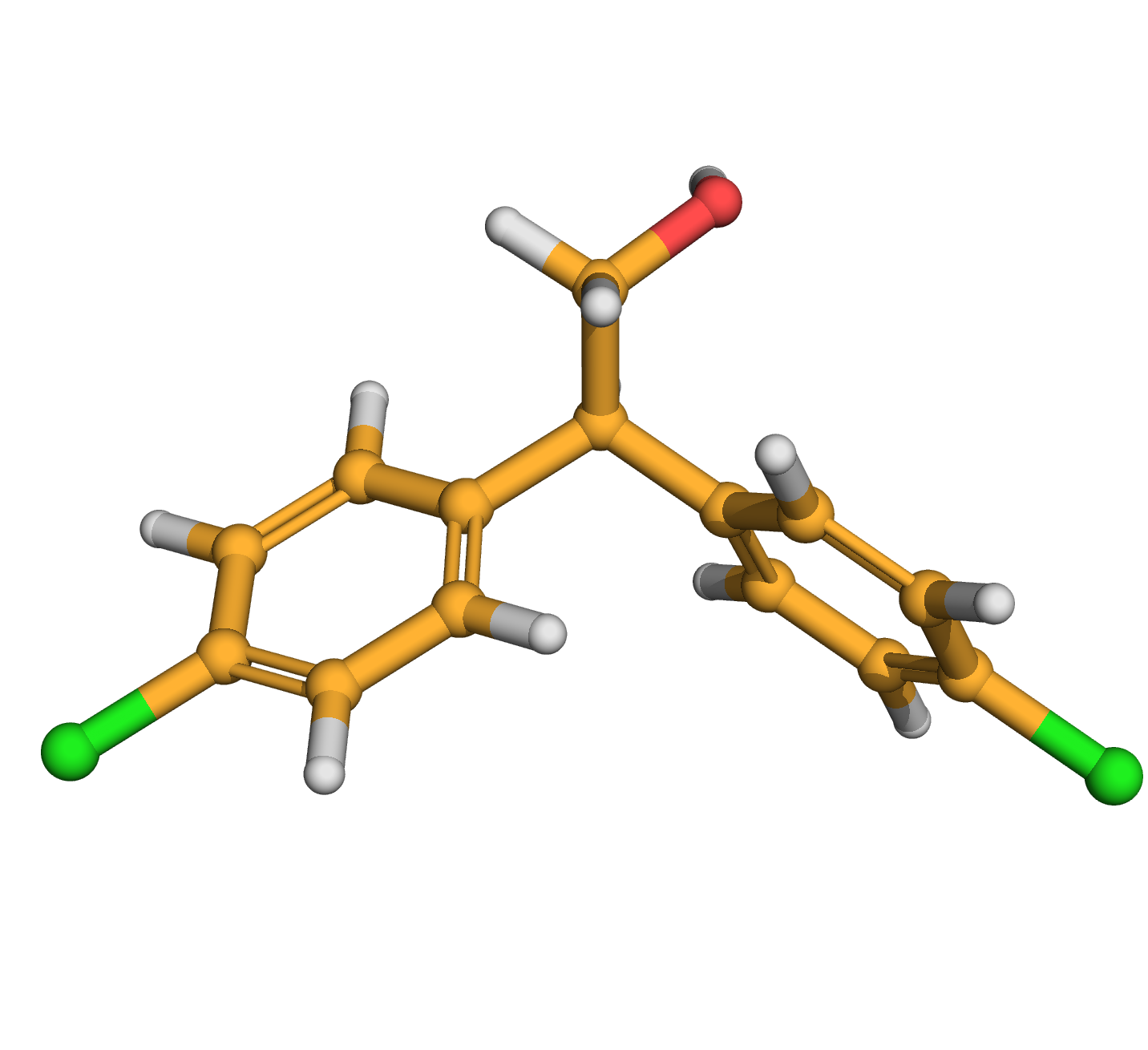
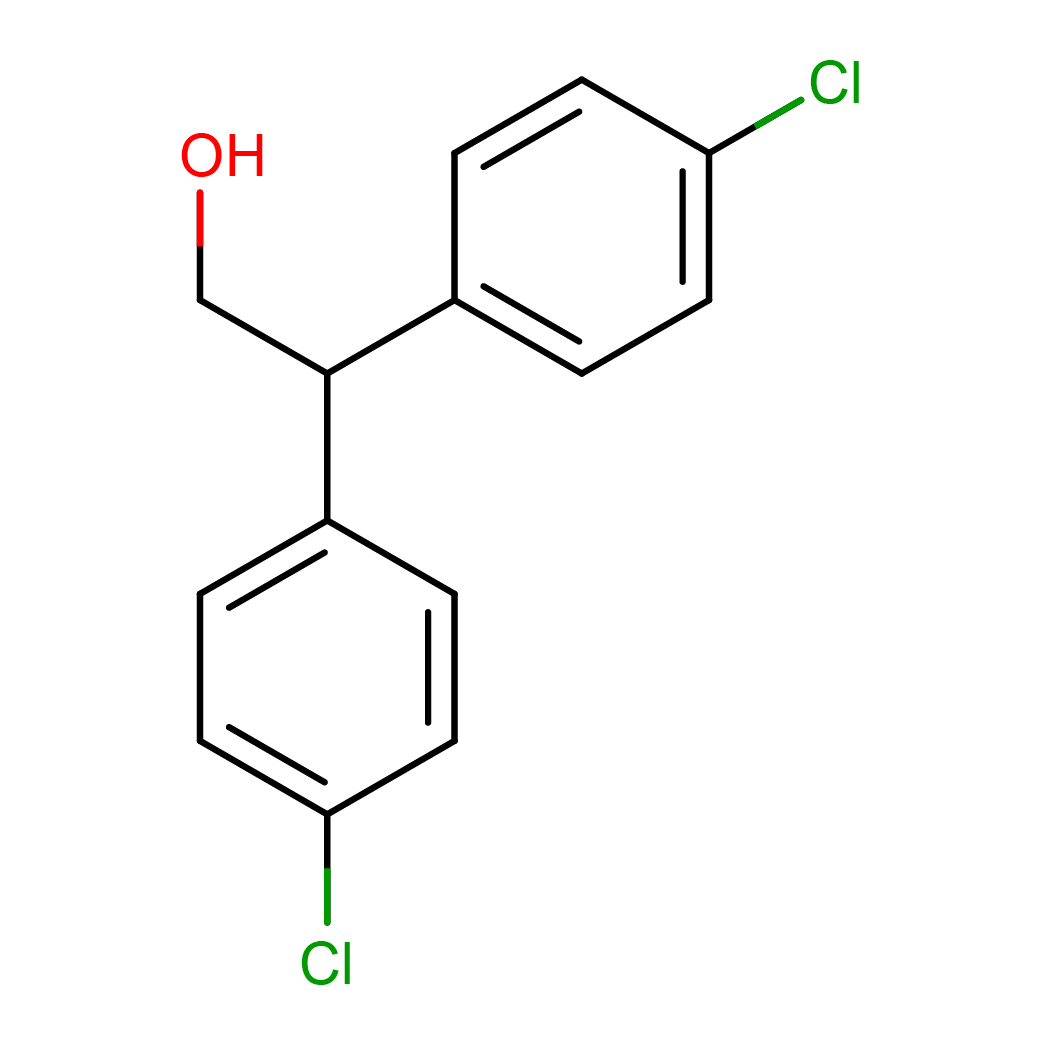
DDOH
Synonyms: "2,2-bis(4-chlorophenyl)ethanol", "2,2-bis(p-chlorophenyl)ethanol", "DDOM", "p,p'-Ddoh"
Source: 2,2-bis(4-chlorophenyl)ethanol is a metabolite of the insecticide DDT.
Identifiers:
IUPAC Name: 2,2-bis(4-chlorophenyl)ethanol
CAS Number: 2642-82-2
PubChem ID: 17533
InChiKey: ZVIDYKRNLNAXFT-UHFFFAOYSA-N
Canonical SMILES: C1=CC(=CC=C1C(CO)C2=CC=C(C=C2)Cl)Cl
Structural Properties:
Molecular Formula: C14H12Cl2O
Molecular Weight: 267.150
Pharmacophore Features:
Number of bond donors: 1
Number of bond acceptors: 1
Number of atoms different from hydrogen: 17
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Cheek AO, Kow K, Chen J, McLachlan JA. 1999. Potential mechanisms of thyroid disruption in humans: Interaction of organochlorine compounds with thyroid receptor, transthyretin, and thyroid-binding globulin. Environ Health Perspect 107(4):273-278.
Vonier PM, Crain DA, McLachlan JA, Guillette LJ Jr., Arnold SF. 1996. Interaction of environmental chemicals with the estrogen and progesterone receptors from the oviduct of the American alligator. Environ Health Perspect 104(12):1318-1322.
External Links

2D-structure
3D-structure