vinclozolin
Synonyms: "vinclozoline", "ronilan", "ornalin", "vinchlozoline", "vorlan"
Source: vinclozolin is a non-systemic dicarboximide fungicide that is used both pre and post-harvest on fruits, vegetables and ornamental plants for the control of several species of fungi.
Identifiers:
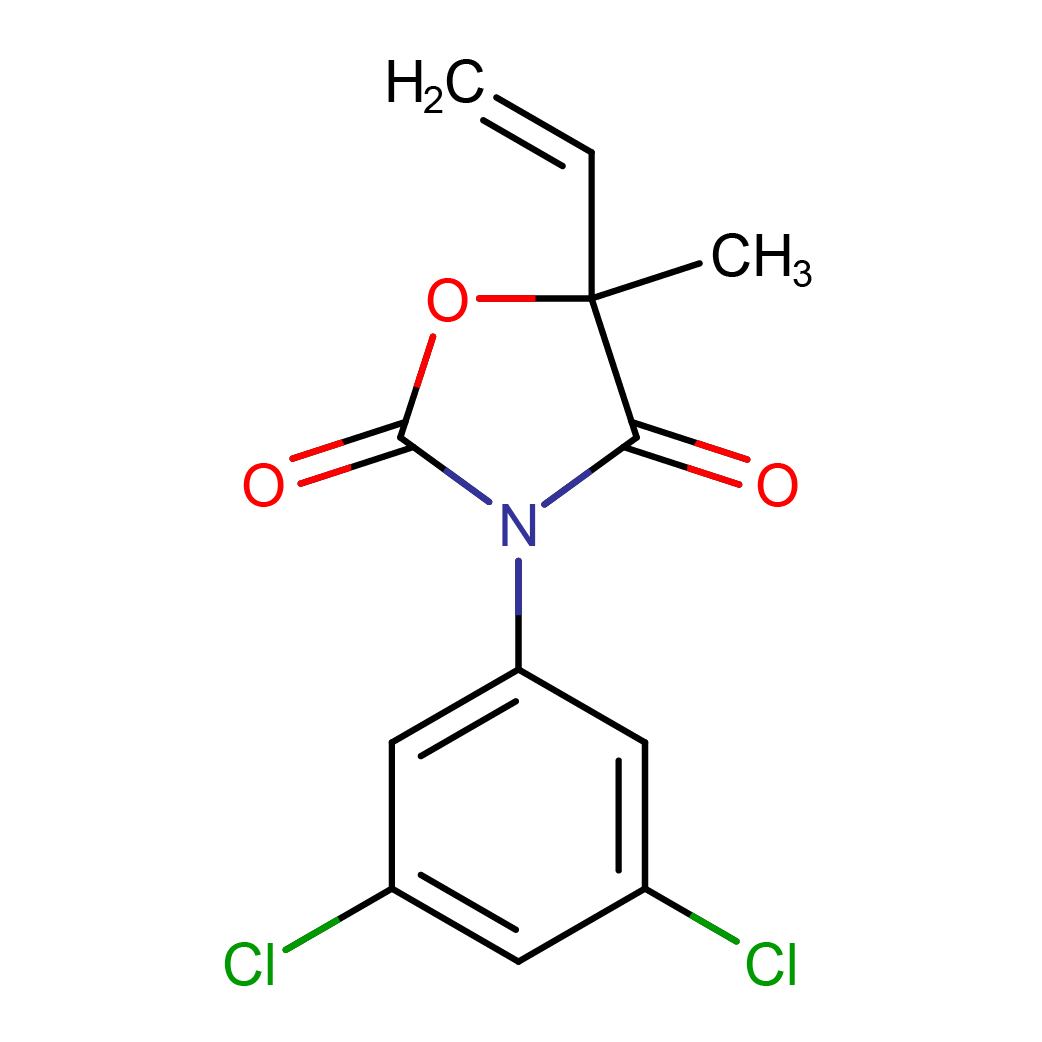
IUPAC Name: 3-(3,5-dichlorophenyl)-5-ethenyl-5-methyl-1,3-oxazolidine-2,4-dione
CAS Number: 50471-44-8
PubChem ID: 39676
InChiKey: FSCWZHGZWWDELK-UHFFFAOYSA-N
Canonical SMILES: CC1(C(=O)N(C(=O)O1)C2=CC(=CC(=C2)Cl)Cl)C=C
Structural Properties:
Molecular Formula: C12H9Cl2NO3
Molecular Weight: 286.111
Pharmacophore Features:
Number of bond donors: 0
Number of bond acceptors: 3
Number of atoms different from hydrogen: 18
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Anway MD, Cupp AS, Uzumcu M, Skinner MK. 2005. Epigenetic transgenerational actions of endocrine disruptors and male fertility. Science 308(5727):1466-1469.
Gray LE Jr., Ostby JS, Kelce WR. 1994. Developmental effects of an environmental antiandrogen - The fungicide vinclozolin alters sex differentiation of the male rat. Toxicol Appl Pharmacol 129(1):46-52.
Kelce WR, Lambright LR, Gray LE, Roberts KP. 1997. Vinclozolin and p,p'-DDE alter androgen-dependent gene expression: In vivo confirmation of an androgen receptor-mediated mechanism. Toxicol Appl Pharmacol 142(1):192-200.Sanderson JT, Boerma J, Lansbergen GW, van den Berg M. 2002. Induction and inhibition of aromatase (CYP19) activity by various classes of pesticides in H295R human adrenocortical carcinoma cells. Toxicol Appl Pharmacol 182(1):44-54.Scippo ML, Argiris C, Van De Weerdt C, Muller M, Willemsen P, Martial J, Maghuin-Rogister G. 2004. Recombinant human estrogen, androgen and progesterone receptors for detection of potential endocrine disruptors. Anal Bioanal Chem 378(3):664-669.
External Links


2D-structure
3D-structure