trifluralin
Synonyms: "treflan", "agreflan", "crisalin", "crisalina", "triflurex", "tristar", "nitran"
Source: trifluralin is a selective, preemergence dinitroaniline herbicide used to control many annual grasses and broadleaf weeds in a large variety of tree fruit, nut, vegetable, and grain crops.
Identifiers:
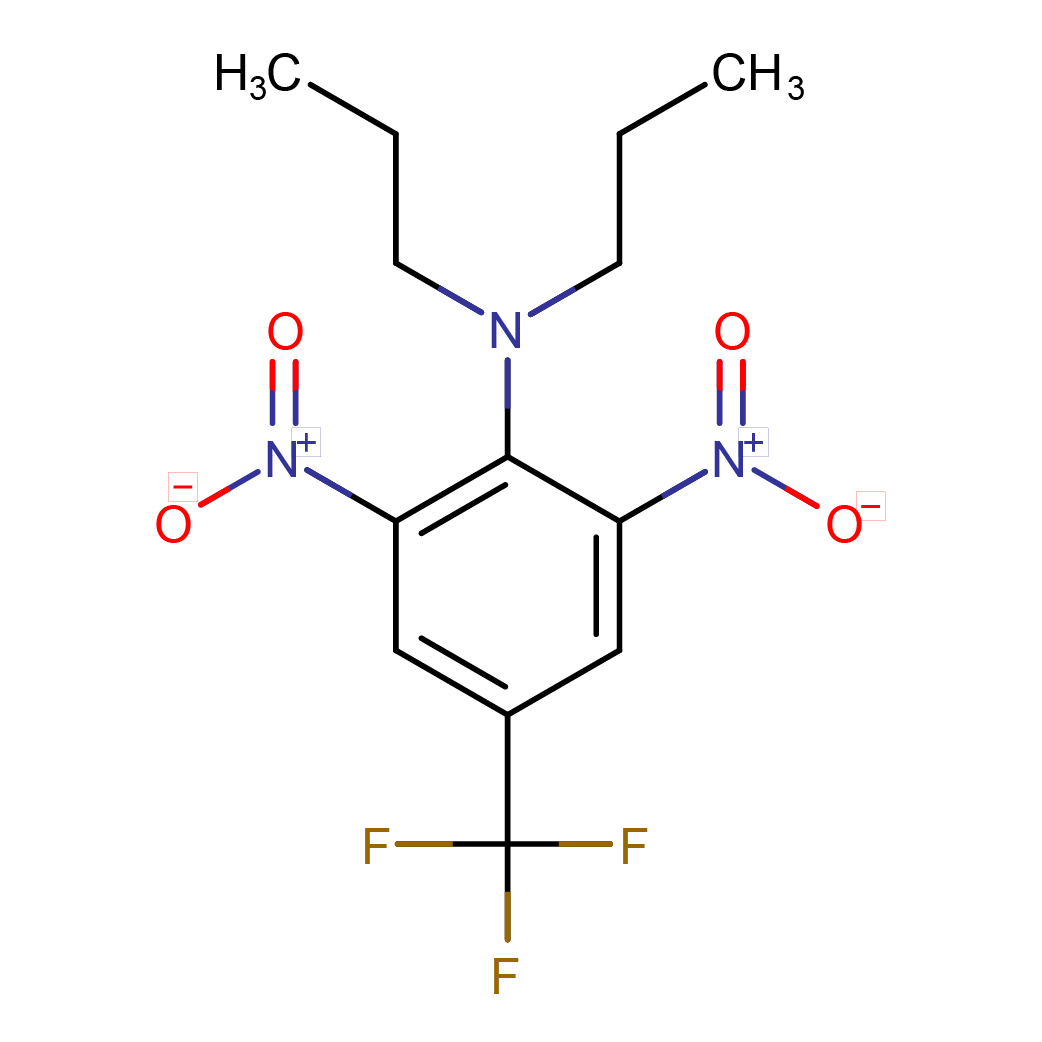
IUPAC Name: 2,6-dinitro-N,N-dipropyl-4-(trifluoromethyl)aniline
CAS Number: 1582-09-8
PubChem ID: 5569
InChiKey: ZSDSQXJSNMTJDA-UHFFFAOYSA-N
Canonical SMILES: CCCN(CCC)C1=C(C=C(C=C1[N+](=O)[O-])C(F)(F)F)[N+](=O)[O-]
Structural Properties:
Molecular Formula: C13H16F3N3O4
Molecular Weight: 335.279
Pharmacophore Features:
Number of bond donors: 0
Number of bond acceptors: 5
Number of atoms different from hydrogen: 23
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Couch JA. 1984. Histopathology and enlargement of the pituitary of a teleost exposed to the herbicide trifluralin. Journal of Fish Diseases 7(2):157-163.
Hurley PM, Hill RN, Whiting RJ. 1998. Mode of carcinogenic action of pesticides inducing thyroid follicular cell tumors in rodents [review]. Environ Health Perspect 106(8):437-445.
Orton F, Lutz I, Kloas W, Routledge EJ. 2009. Endocrine disrupting effects of herbicides and pentachlorophenol: in vitro and in vivo evidence. Environ Sci Technol 43(6):2144-2150.
External Links


2D-structure
3D-structure