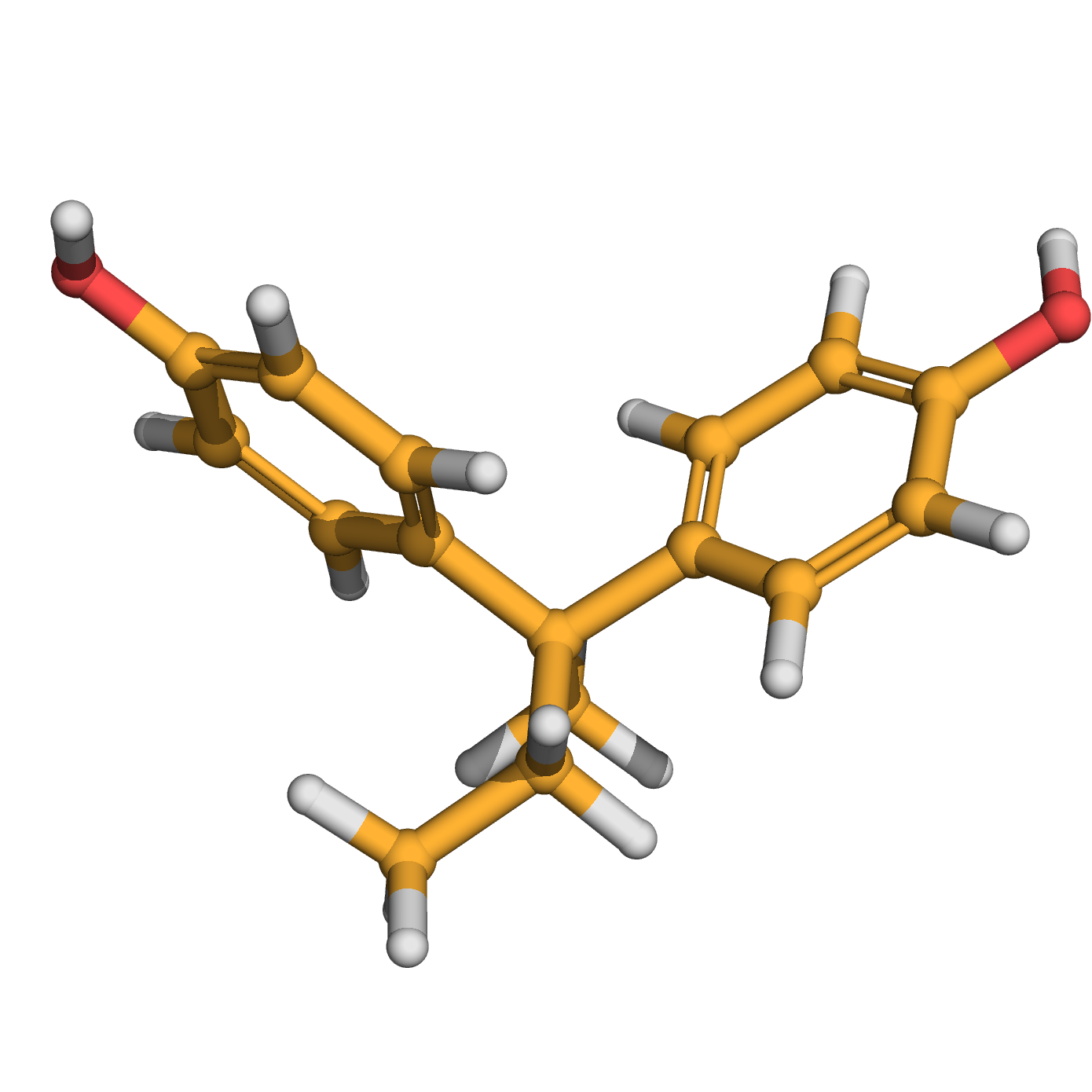
bisphenol B
Synonyms: "2,2-bis(4-hydroxyphenyl)butane", "p,p'-sec-butylidenediphenol", "4,4'-(1-methylpropylidene)bisphenol"
Source: bisphenol B, is the analouge of Bisphenol A, which is used to make certain plastic an epoxy resins.
Identifiers:
IUPAC Name: 4-[2-(4-hydroxyphenyl)butan-2-yl]phenol
CAS Number: 77-40-7
PubChem ID: 66166
InChiKey: HTVITOHKHWFJKO-UHFFFAOYSA-N
Canonical SMILES: CCC(C)(C1=CC=C(C=C1)O)C2=CC=C(C=C2)O
Structural Properties:
Molecular Formula: C16H18O2
Molecular Weight: 242.313
Pharmacophore Features:
Number of bond donors: 2
Number of bond acceptors: 2
Number of atoms different from hydrogen: 18
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Kitamura S, Suzuki T, Sanoh S, Kohta R, Jinno N, Sugihara K, Yoshihara S, Fujimoto N, Watanabe H, Ohta S. 2005. Comparative study of the endocrine-disrupting activity of bisphenol A and 19 related compounds. Toxicol Sci 84(2):249-259.
Nishihara T, Nishikawa J, Kanayama T, Dakeyama F, Saito K, Imagawa M, Takatori S, Kitagawa Y, Hori S, Utsumi H. 2000. Estrogenic activities of 517 chemicals by yeast two-hybrid assay. Journal of Health Science 46(4):282-298.
Terasaka S, Inoue A, Tanji M, Kiyama R. 2006. Expression profiling of estrogen-responsive genes in breast cancer cells treated with alkylphenols, chlorinated phenols, parabens, or bis- and benzoylphenols for evaluation of estrogenic activity. Toxicol Lett 163(2):130-141.
Yamasaki K, Takeyoshi M, Sawaki M, Imatanaka N, Shinoda K, Takatsuki M. 2003. Immature rat uterotrophic assay of 18 chemicals and Hershberger assay of 30 chemicals. Toxicology 183(1-3):93-115.
Yoshihara S, Makishima M, Suzuki N, Ohta S. 2001. Metabolic activation of bisphenol A by rat liver S9 fraction. Toxicol Sci 62(2):221-227.
External Links


2D-structure
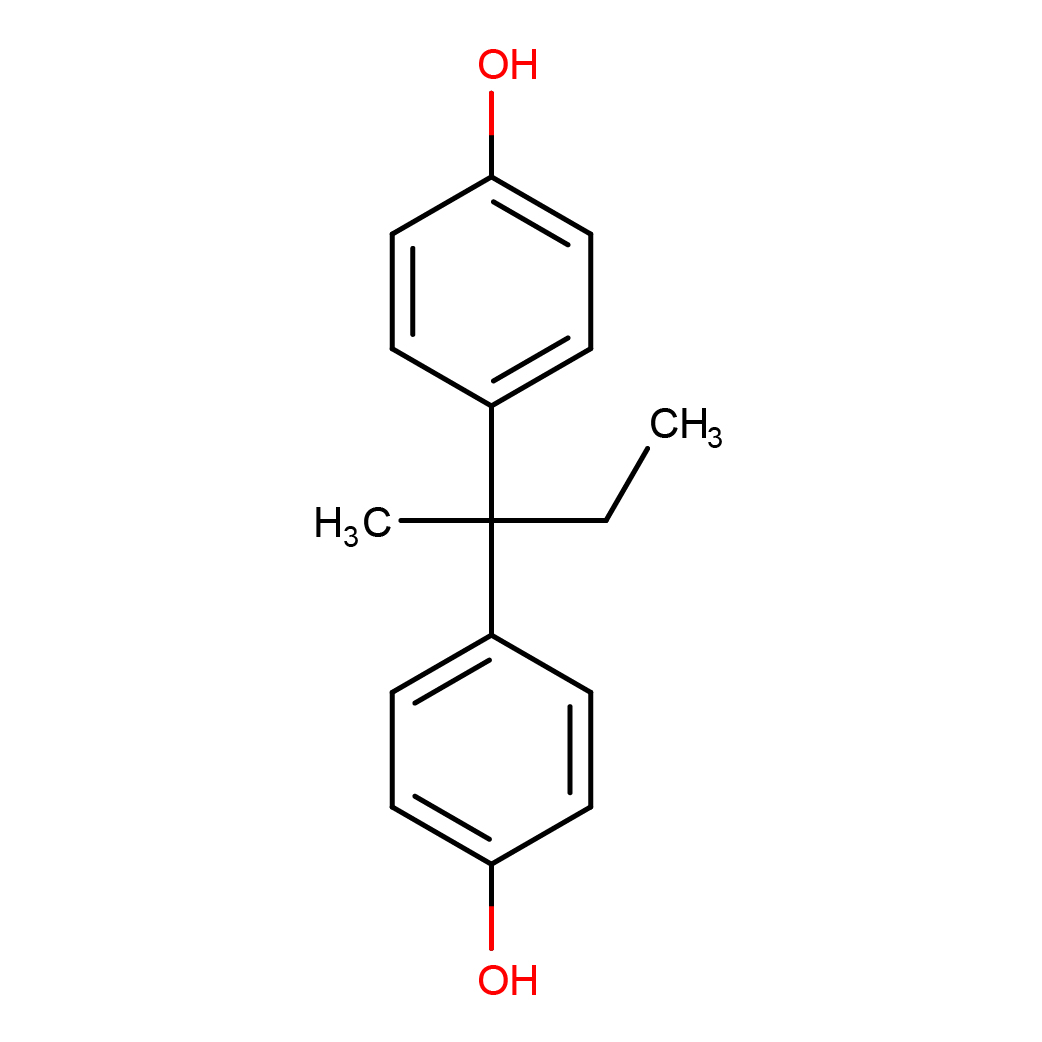
3D-structure