perfluorohexanesulfonic acid
Synonyms: "perfluorohexanesulfonic acid", "355-46-4", "1,1,2,2,3,3,4,4,5,5,6,6,6-tridecafluorohexane-1-sulfonic acid", "perfluorohexane sulfonic acid", "perfluorohexane-1-sulphonic acid", "PFHxS", "perfluorohexane-1-sulfonic acid", "perfluorohexanesulphonic acid", "tridecafluorohexane-1-sulfonic acid".
Source: perfluorohexanesulfonic acid is used to make other organic chemicals containing fluorine.
Identifiers:
IUPAC Name: 1,1,2,2,3,3,4,4,5,5,6,6,6-tridecafluorohexane-1-sulfonic acid
CAS Number: 355-46-4
PubChem ID: 67734
InChiKey: QZHDEAJFRJCDMF-UHFFFAOYSA-N
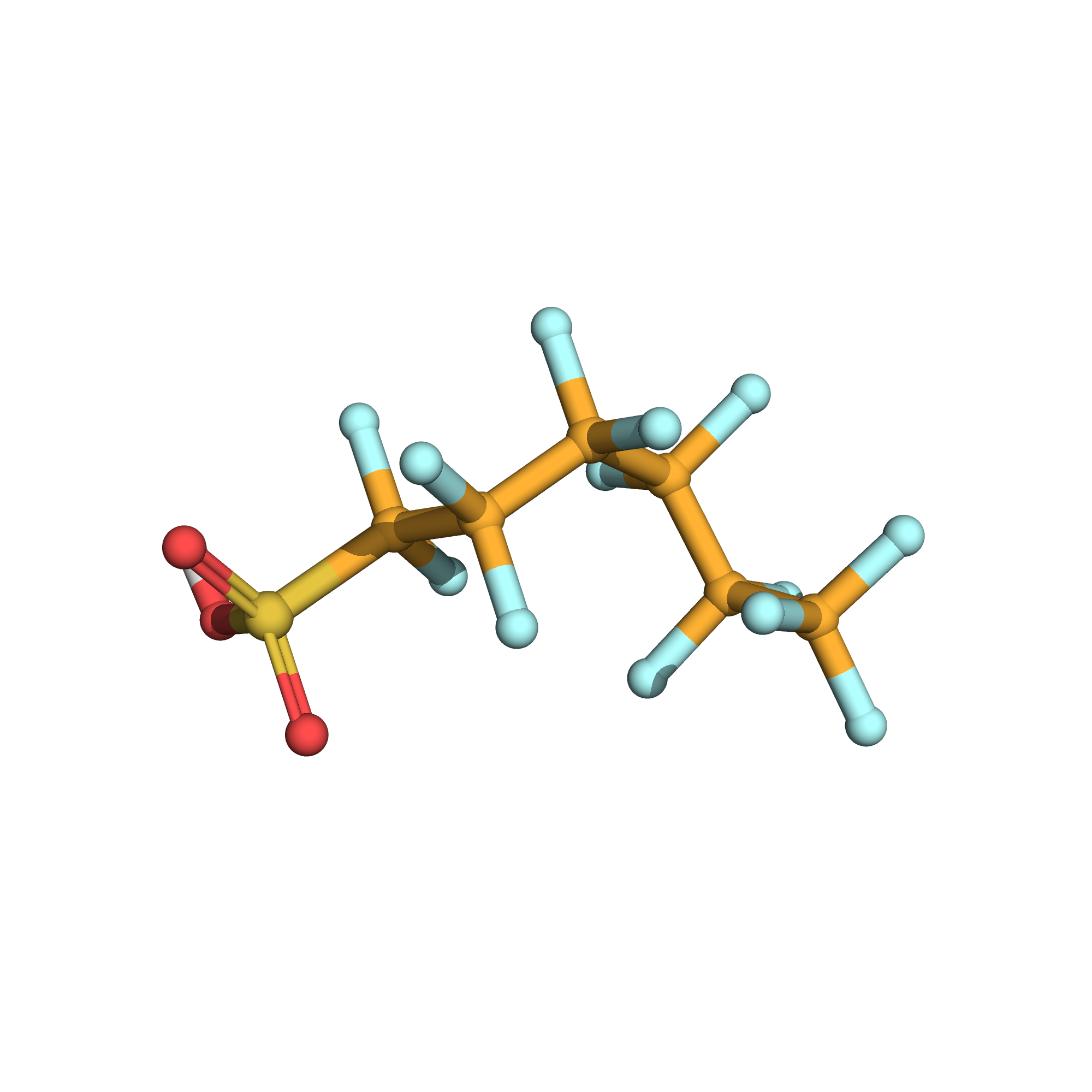
Canonical SMILES: C(C(C(C(F)(F)S(=O)(=O)O)(F)F)(F)F)(C(C(F)(F)F)(F)F)(F)F
Structural Properties:
Molecular Formula: C6HF13O3S
Molecular Weight: 400.110
Pharmacophore Features:
Number of bond donors: 1
Number of bond acceptors: 16
Number of atoms different from hydrogen: 23
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Cassone CG, Vongphachan V, Chiu S, Williams, KL, Letcher RJ, Pelletier E, Crump D and Kennedy SW. 2012. In ovo effects of perfluorohexane sulfonate and perfluorohexanoate on pipping success, development, mRNA expression, and thyroid hormone levels in chicken embryos. Toxicological Sciences 127(1):216-224. DOI: 10.1093/toxsci/kfs072. URL: https://www.ncbi.nlm.nih.gov/pubmed/22302310.
Kjeldsen LS and Bonefeld-Jargensen EC. 2013. Perfluorinated compounds affect the function of sex hormone receptors. Environ Sci Pollut Res Int 20(11):8031-8044. DOI: 10.1007/s11356-013-1753-3. URL: https://www.ncbi.nlm.nih.gov/pubmed/23764977.
Long M, Ghisari M and Bonefeld-Jargensen EC. 2013. Effects of perfluoroalkyl acids on the function of the thyroid hormone and the aryl hydrocarbon receptor. Environ Sci Pollut Res Int 20(11):8045-8056. DOI: 10.1007/s11356-013-1628-7. URL: https://www.ncbi.nlm.nih.gov/pubmed/23539207.
Maisonet M, Calafat AM, Marcus M, Jaakkola JJ and Lashen H. 2015. Prenatal exposure to perfluoroalkyl acids and serum testosterone concentrations at 15 years of age in female ALSPAC study participants. Environ Health Perspect 123(12):1325-1330. DOI: 10.1289/ehp.1408847. URL: https://ehp.niehs.nih.gov/1408847/.
Rosen MB, Das KP, Rooney J, Abbott B, Lau C and Corton JC. 2017. PPAR?-independent transcriptional targets of perfluoroalkyl acids revealed by transcript profiling. Toxicology 387:95-107. DOI: 10.1016/j.tox.2017.05.013. URL: https://www.ncbi.nlm.nih.gov/pubmed/28558994.
Rosenmai AK, Ahrens L, Godec T, Lundqvist J and Oskarsson A. 2017. Relationship between peroxisome proliferator?activated receptor alpha activity and cellular concentration of 14 perfluoroalkyl substances in HepG2 cells. J Appl Toxicol 38(2):219-226. DOI: 10.1002/jat.3515. URL: https://www.ncbi.nlm.nih.gov/pubmed/28857218.
Shah-Kulkarni, S., Kim, B.M., Hong, Y.C., Kim, H.S., Kwon, E.J., Park, H., Kim, Y.J. and Ha, E.H., 2016. Prenatal exposure to perfluorinated compounds affects thyroid hormone levels in newborn girls. Environment international, 94, pp.607-613. DOI: 10.1016/j.envint.2016.06.024. URL: http://www.sciencedirect.com/science/article/pii/S0160412016302422.
Vongphachan V, Cassone CG, Wu D, Chiu S, Crump D, Kennedy SW. 2011. Effects of perfluoroalkyl compounds on mRNA expression levels of thyroid hormone-responsive genes in primary cultures of avian neuronal cells. Toxicol Sci 120(2):392-402. DOI: 10.1093/toxsci/kfq395. URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3061477/Watkins AM, Wood CR, Lin MT and Abbott BD. 2015. The effects of perfluorinated chemicals on adipocyte differentiation in vitro. Mol Cell Endocrinol 400:90-101. DOI: 10.1016/j.mce.2014.10.020. URL: https://www.ncbi.nlm.nih.gov/pubmed/25448844.
Zhao B, Lian Q, Chu Y, Hardy DO, Li XK and Ge RS. 2011. The inhibition of human and rat 11?-hydroxysteroid dehydrogenase 2 by perfluoroalkylated substances. The J Steroid Biochem Mol Biol 125(1-2):143-147. DOI: 10.1016/j.jsbmb.2010.12.017. URL: https://www.ncbi.nlm.nih.gov/pubmed/21237268.
External Links


2D-structure
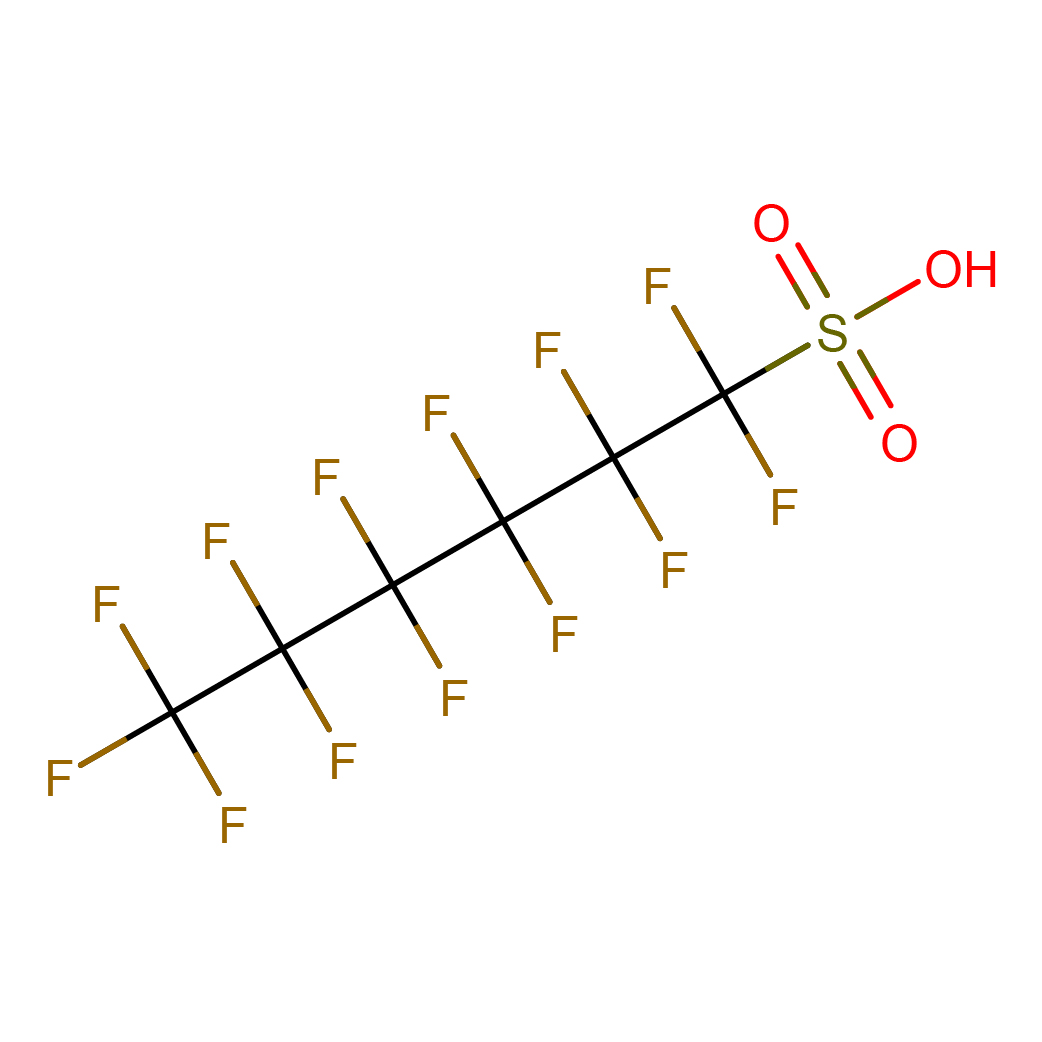
3D-structure