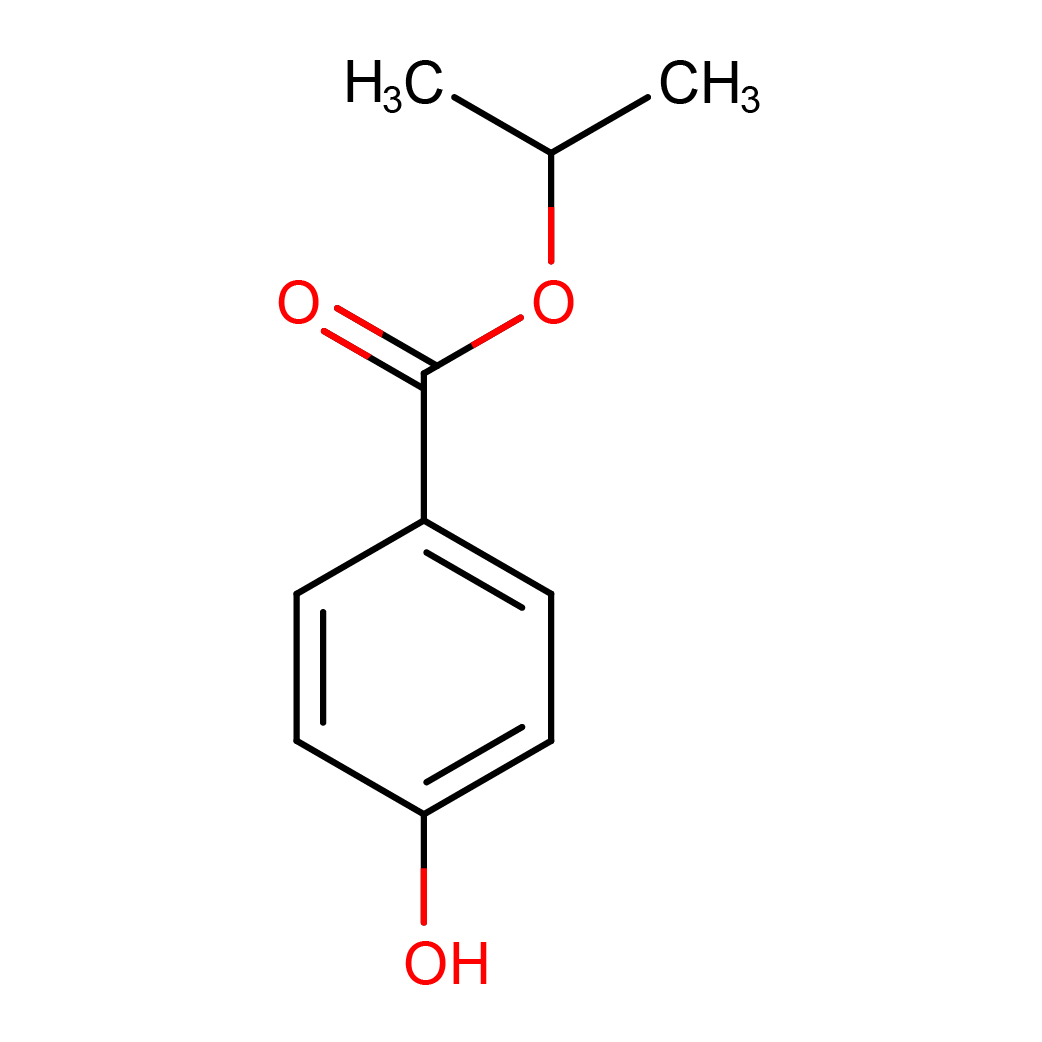
isopropyl 4-hydroxybenzoate
Synonyms: "Isopropyl 4-hydroxybenzoate", "isopropylparaben", "isopropyl p-hydroxybenzoate", "isopropyl paraben", "propan-2-yl 4-hydroxybenzoate", "p-hydroxybenzoic acid isopropyl ester", "isopropylhydroxybenzoate", "4-hydroxybenzoic acid isopropyl ester", "p-oxybenzoesaureisopropylester", "1-methylethyl 4-hydroxybenzoate", "isopropyl-4-hydroxybenzoate".
Source: Isopropylparaben is in the parabens family of preservatives used by the food, pharmaceutical, and personal care products.
Identifiers:
IUPAC Name: propan-2-yl 4-hydroxybenzoate
CAS Number: 4191-73-5
PubChem ID: 20161
InChiKey: CMHMMKSPYOOVGI-UHFFFAOYSA-N
Canonical SMILES: CC(C)OC(=O)C1=CC=C(C=C1)O
Structural Properties:
Molecular Formula: C10H12O3
Molecular Weight: 180.203
Pharmacophore Features:
Number of bond donors: 1
Number of bond acceptors: 3
Number of atoms different from hydrogen: 13
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Endocrine Disruptor
TEDX List of Potential Endocrine Disruptors

Okubo T, Yokoyama Y, Kano K, Kano I. 2001. ER-dependent estrogenic activity of parabens assessed by proliferation of human breast cancer MCF-7 cells and expression of ER[alpha] and PR. Food Chem Toxicol 39(12):1225-1232. DOI: 10.1016/S0278-6915(01)00073-4. URL: https://www.sciencedirect.com/science/article/pii/S0278691501000734?via%3Dihub.
Terasaki M, Kamata R, Shiraishi F, Makino M. 2009. Evaluation of estrogenic activity of parabens and their chlorinated derivatives by using the yeast two-hybrid assay and the enzyme-linked immunosorbent assay. Environ Toxicol Chem 28(1):204-208. DOI: 10.1897/08-225.1. URL: http://onlinelibrary.wiley.com/doi/10.1897/08-225.1/full.
Vo TT, Jeung EB. 2009. An evaluation of estrogenic activity of parabens using uterine calbindin-d9k gene in an immature rat model. Toxicol Sci 112(1):68-77. DOI: 10.1093/toxsci/kfp176. URL: https://academic.oup.com/toxsci/article/112/1/68/1636403.
External Links




2D-structure
3D-structure